How Conquering Cancer Is a Team Effort

After finishing her academic studies, Dr. Mittendorf enlisted in the U.S. Air Force—an experience that would propel her into the field of oncology.
AI Avatar–Based Education Leads to Improved Patient Understanding of Radiation Treatment Plans

A new study has shown that AI avatar–based digital patient engagement prior to in-person radiation treatment consultations may enable patients to feel more knowledgeable and less stressed than patients who did not engage with an AI avatar, according to findings presented during the Congress of the European Society for Radiotherapy and Oncology (ESTRO 2026; Abstract 4187).
ASCENDE-RT 15-Year Update Shows No Overall Survival Benefit, With ‘Borderline’ Signal for Fewer Deaths

Fifteen-year results from the phase III ASCENDE-RT trial in patients with intermediate- and high-risk localized prostate cancer receiving androgen-deprivation therapy (ADT) and pelvic external-beam radiotherapy (EBRT) show no significant overall survival advantage with a prostate brachytherapy boost vs an EBRT boost, with a signal toward improved prostate cancer–specific survival that was sensitive to challenges in determining cause of death, according to findings presented by Scott Tyldesley, MD, FRCPC, of BC Cancer Vancouver, at the 2026 ASCO Genitourinary (GU) Cancers Symposium.1
FDA Approves Atezolizumab for Adjuvant Treatment of MRD-Positive Muscle-Invasive Bladder Cancer
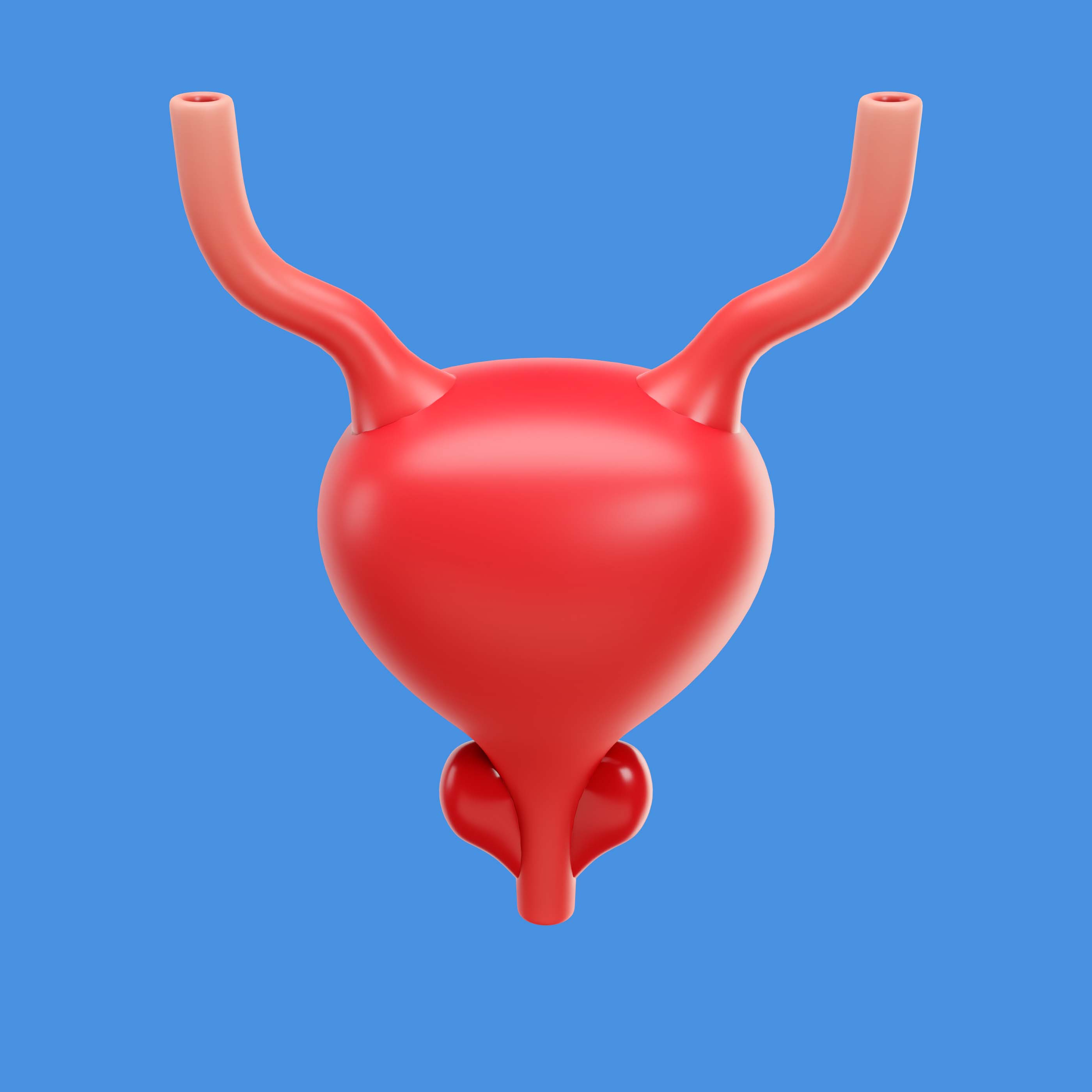
On May 15, the U.S. Food and Drug Administration (FDA) approved the PD-L1 inhbitor atezolizumab (Tecentriq) and atezolizumab and hyaluronidase-tqjs (Tecentriq Hybreza) as adjuvant therapies for adults with muscle-invasive bladder cancer (MIBC) after cystectomy who have circulating tumor DNA molecular residual disease (ctDNA MRD), as determined by an FDA-authorized test.
Relacorilant Plus Nab-Paclitaxel Improves Overall Survival in Platinum-Resistant Ovarian Cancer

Final overall survival results from the phase III ROSELLA trial showed a statistically and clinically significant survival benefit with the selective glucocorticoid receptor antagonist relacorilant plus nab-paclitaxel vs nab-paclitaxel alone in patients with platinum-resistant ovarian cancer. Median overall survival was 16.0 months with the combination compared with 11.9 months with nab-paclitaxel monotherapy, yielding a hazard ratio (HR) of 0.65 and a 35% reduction in the risk of death.
Sponsored Content

SPONSORED BY LILLY MEDICAL AFFAIRS
Treatment Considerations for Relapsed/Refractory CLL in the Post-cBTKi SettingMore Top Stories
I’m Young and Have Advanced Neuroendocrine Carcinoma of the Rectum
Eight years ago, I was 33 years old, and my main health concern was a diagnosis of ankylosing spondylitis, a form of arthritis that causes stiff, painful joints in the spine. Having a chronic disease made me pay close attention to any changes in my health, so when I noticed blood in my stool, I...
Head-to-Head on Headspace: Cognitive Effects of Darolutamide vs Enzalutamide in Advanced Prostate Cancer
Patients with advanced prostate cancer treated with darolutamide experienced significantly less objectively assessed cognitive decline over 24 weeks than those receiving enzalutamide, according to results from the prospective, randomized open-label phase II ARACOG (AFT-47) trial to be presented at...
New ASCO Guideline Fills Gap in Guidance on Rapidly Evolving Treatment Options in Thyroid Cancer
ASCO has issued a new clinical practice guideline on the use of systemic therapy for treatment of different types of thyroid cancer, a field that has changed substantially in recent years.1 “Despite a rather rapidly evolving field of targeted and nontargeted systemic agents in the management of...
Patient Care Is Not What We Do, But What Patients Perceive
MUHAMMAD RAFIQUL ISLAM, MBBS, MD, MSPH, FACP In modern health care, patient care is often defined by clinical actions such as diagnoses made, treatments delivered, and protocols followed. Clinicians are trained to prioritize technical accuracy, evidence-based interventions, and measurable...
GLP-1 RAs May Reduce Metastatic Progression in Certain Obesity-Related Cancers
Real-world data suggest that GLP-1 receptor agonists (RAs) may reduce metastatic progression in certain obesity-related cancers, particularly lung, breast, colorectal, and liver cancers, according to findings to be presented at the 2026 ASCO Annual Meeting (Abstract 3143). GLP-1 receptor expression ...
Gerald Hsu, MD, PhD, Named Editor-in-Chief of the ASCO Educational Book
ASCO is pleased to announce the appointment of Gerald Hsu, MD, PhD, as the new Editor-in-Chief of the ASCO Educational Book. Dr. Hsu is a Professor of Clinical Medicine at the University of California, San Francisco (UCSF), where he has served in numerous educational leadership roles. Since 2018,...
Yoga Intervention May Reduce Mood Disturbance, Anxiety, Fatigue, and Insomnia in Cancer Survivors
A 4-week structured yoga intervention improved overall mood disturbance, anxiety, and fatigue among cancer survivors, according to findings from a phase III randomized controlled trial that will be presented at the 2026 ASCO Annual Meeting (Abstract 12004). The analysis also suggested that...








