Robin Cornelissen, MD, PhD, on HER2 Exon 20–Mutated NSCLC: Poziotinib in Treatment-Naive Disease
ESMO Congress 2021
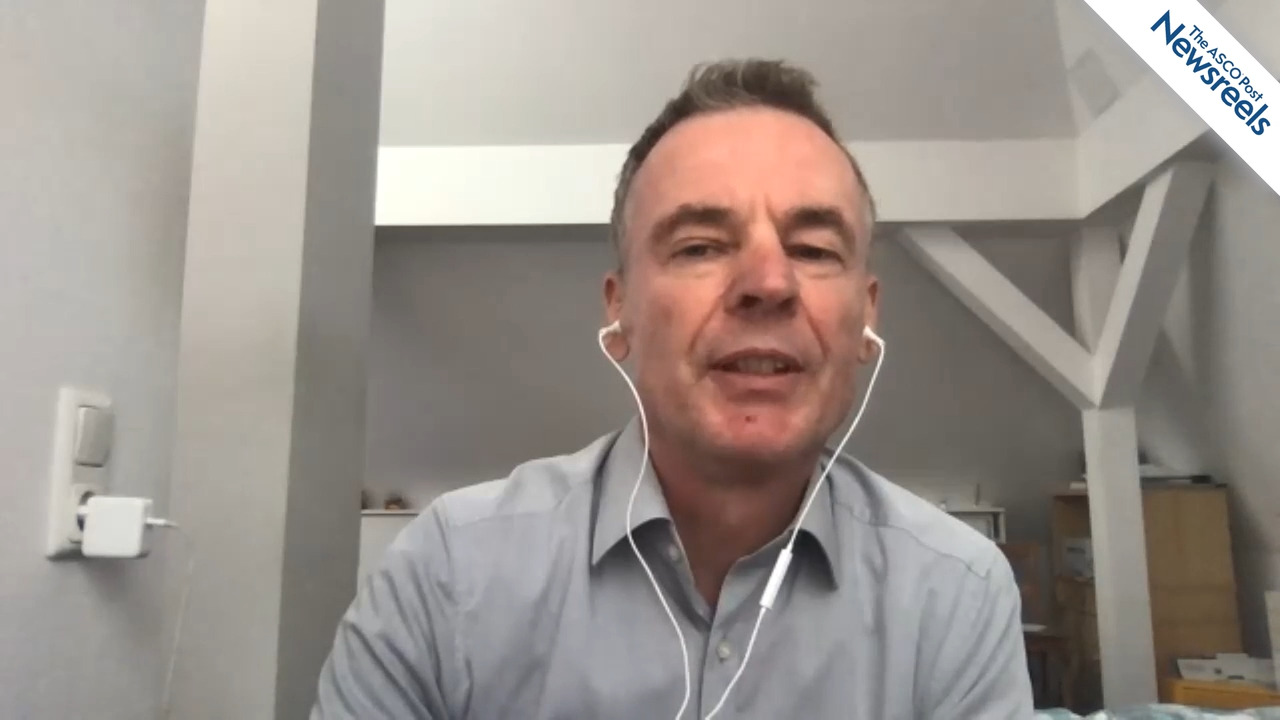
Robin Cornelissen, MD, PhD, of Erasmus University in Rotterdam, discusses phase II findings from the ZENITH20-4 study, which explored the question of whether poziotinib could benefit patients whose newly diagnosed non–small cell lung cancer harbors EGFR and HER2 exon 20 mutations. Potentially, this novel tyrosine kinase inhibitor may fill an unmet medical need (Abstract LBA46).
The ASCO Post Staff
Dieter Hörsch, MD, of Germany’s Central Clinic in Bad Berka, discusses phase III results from the SPINET trial, the largest prospective study to date of the somatostatin analog lanreotide autogel. The study suggests that this agent may prove to be an appropriate treatment option for patients with somatostatin receptor–positive bronchopulmonary neuroendocrine tumors, especially typical carcinoids (Abstract 1096O).
The ASCO Post Staff
Javier Cortés, MD, PhD, of Barcelona’s IOB Institute of Oncology, discusses phase III data from the DESTINY-Breast03 study, which support trastuzumab deruxtecan becoming the standard of care for second-line treatment of women with HER2-positive metastatic breast cancer (Abstract LBA1).
The ASCO Post Staff
Naveen S. Vasudev, PhD, MBChB, of the University of Leeds, discusses phase II results from the PRISM trial, which showed that giving ipilimumab every 12 weeks instead of every 3 weeks, in combination with nivolumab, led to lower rates of grade 3 and 4 toxicities in patients with advanced renal cell carcinoma. Efficacy appeared to be comparable between both arms (Abstract LBA29).
The ASCO Post Staff
Toni K. Choueiri, MD, of Dana-Farber Cancer Institute, discusses patient-reported outcomes for quality of life in the KEYNOTE-564 study, which previously met its primary endpoint of disease-free survival with adjuvant pembrolizumab vs placebo following surgery for renal cell carcinoma (Abstract 653O).
The ASCO Post Staff
Jason J. Luke, MD, of UPMC Hillman Cancer Center, discusses phase III results showing that adjuvant pembrolizumab for patients with resected stage IIB and IIC melanoma decreased the risk of disease recurrence or death by 35% compared with placebo. It was also associated with significantly prolonged recurrence-free survival (Abstract LBA3).