Helena M. Earl, MBBS, PhD, on HER2-Positive Early Breast Cancer: A Meta-analysis of Trastuzumab Trials
ESMO Congress 2021
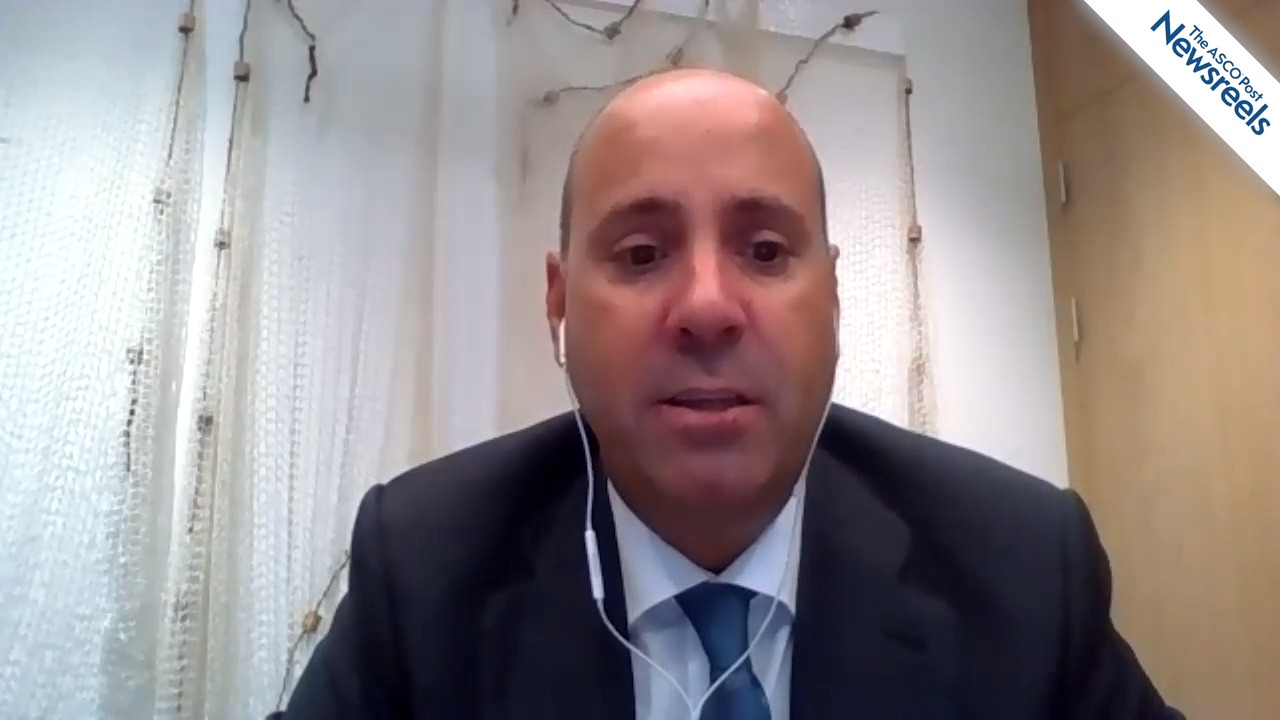
Helena M. Earl, MBBS, PhD, of the University of Cambridge, discusses an individual patient data meta-analysis of noninferiority randomized clinical trials to determine whether a duration of less than the standard of 12 months of adjuvant trastuzumab is noninferior for treatment outcomes in patients with HER2-positive early breast cancer (Abstract LBA11).
The ASCO Post Staff
Javier Cortés, MD, PhD, of Barcelona’s IOB Institute of Oncology, discusses phase III data from the DESTINY-Breast03 study, which support trastuzumab deruxtecan becoming the standard of care for second-line treatment of women with HER2-positive metastatic breast cancer (Abstract LBA1).
The ASCO Post Staff
Thomas Powles, MD, PhD, of Queen Mary University of London, discusses phase II results from the NORSE study, which showed that the kinase inhibitor erdafitinib plus the monoclonal antibody cetrelimab produced meaningful responses in cisplatin-ineligible patients with first-line metastatic or locally advanced urothelial carcinoma and fibroblast growth factor receptor (FGFR) alterations (Abstract LBA27).
The ASCO Post Staff
Susana N. Banerjee, MBBS, PhD, of The Royal Marsden NHS Foundation Trust, discusses phase I results that have generated interest in the combination of the RAF/MEK inhibitor VS-6766 and the FAK inhibitor defactinib for patients with recurrent low-grade serous ovarian cancer, a disease that typically has limited response to conventional chemotherapy and hormonal therapy. The data support ongoing investigation (Abstract 725MO).
The ASCO Post Staff
Nicoletta Colombo, MD, of the Istituto Europeo Oncologico, discusses phase III results that showed improvements in progression-free and overall survival with a combination of pembrolizumab plus chemotherapy, compared with placebo and chemotherapy, for patients with persistent, recurrent, or metastatic cervical cancer. These benefits were seen regardless of PD-L1 expression and concomitant bevacizumab use, suggesting that pembrolizumab plus chemotherapy, with or without bevacizumab, may be a new standard of care for this population (Abstract LBA2).
The ASCO Post Staff
Hope S. Rugo, MD, of the University of California, San Francisco, discusses phase III results from the KEYNOTE-355 study of pembrolizumab plus chemotherapy, which improved overall survival vs chemotherapy alone in patients with previously untreated locally recurrent, inoperable, or metastatic triple-negative breast cancer whose tumors expressed PD-L1 (Abstract LBA16).