Rami Manochakian, MD, on NSCLC: Clinical Implications of Findings on Nivolumab Plus Chemotherapy
2022 ASCO Annual Meeting
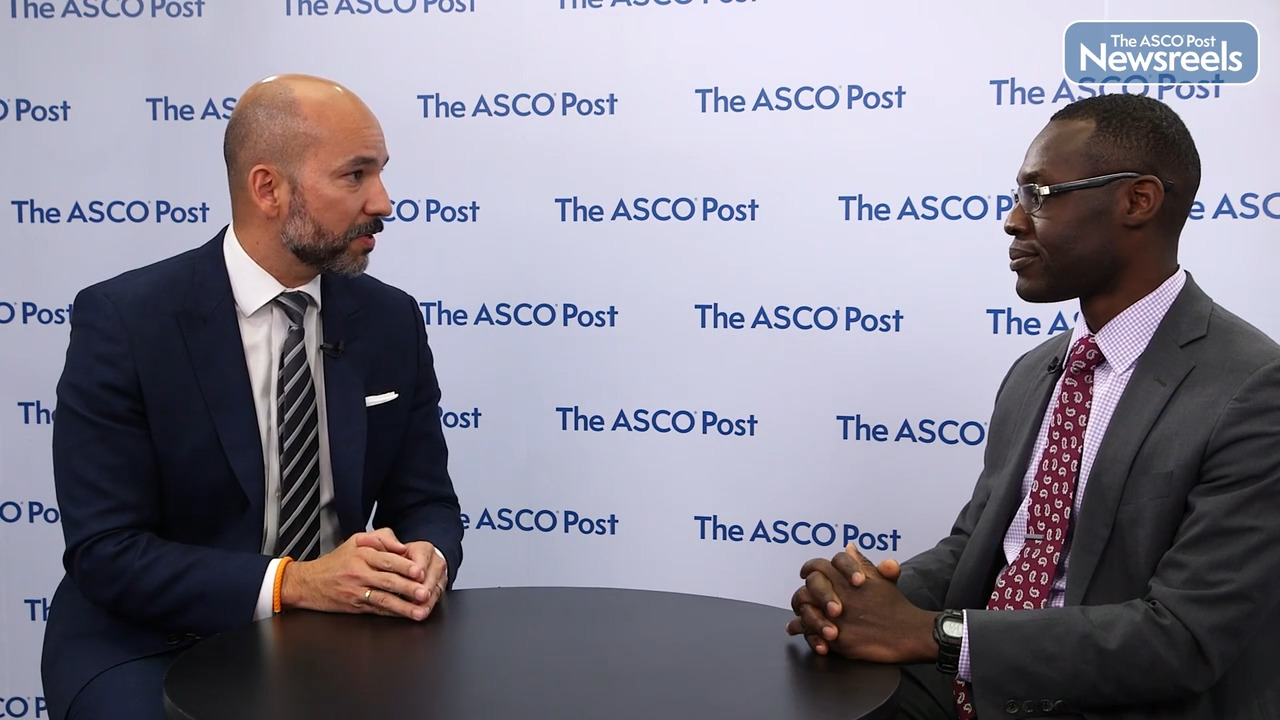
Rami Manochakian, MD, of Mayo Clinic Florida, discusses the phase II findings of the NADIM II trial, which confirmed that, in terms of pathologic complete response as well as the feasibility of surgery, combining nivolumab and chemotherapy was superior to chemotherapy alone as a neoadjuvant treatment for locally advanced, resectable stage IIIA non–small cell lung cancer (Abstract 8501).
Transcript
Disclaimer: This video transcript has not been proofread or edited and may contain errors.
The NADIM II Trial is a randomized open label Phase II trial of Neoadjuvant Nivolumab with a regimen, chemotherapy regimen, of carboplatin paclitaxel versus chemotherapy alone, given in three cycles for patients with Stage 3A non-small cell lung cancer. After the three cycle of the neoadjuvant therapy, patient proceeded with surgery, and following surgery, patient received six months of Adjuvant Nivolumab. This study was done by the Spanish Lung Cancer Group Trial. The study primary endpoint was the pathological complete response rate, and secondary endpoint, there was the major pathological response rate, as well as response rate and also adverse events. This study is important, since it's really looking in particular at the Stage 3A patients with non-small cell lung cancer. This is a challenging population.
There is an evolving research and trials testing in particularly this population. We have recently reported CheckMate 816, that led to the approval of Neoadjuvant Nivolumab and chemotherapy in patients from Stage 1B to Stage 3. We have the Adjuvant therapy also approved in a patient with Stage 3. We have the patient who don't undergo resection, and they receive concurrent chemoradiation. So, what this trial is come as a validation to the CheckMate 816, looking in particularly to this patient of a Stage 3A.
The result of the studies were positive. The primary endpoint, which was the pathological complete response rate, was about 36% versus 7% in the patients who did not get the immunotherapy and received the chemotherapy alone. The secondary endpoint, the major pathological response rate, which means the 10% or less of viable tumor in the resected specimen and lymph node, was 52% versus 13%. The overall response rate was 74% versus 48%. And the adverse event, there was some modest increase in the adverse event, in particularly the Grade 3, 4. It was about 24% versus 20%.
This study is, again, comes as a validation for the role of Neoadjuvant chemotherapy and immunotherapy in patient with Stage 3A. This is something that has continued to evolve, as I mentioned earlier, and it's definitely set a standard of care option as one of the option for patients who potentially have resectable Stage 3 non-small cell lung cancer to receive chemotherapy and immunotherapy, followed by surgery and followed by Adjuvant immunotherapy.
Related Videos
The ASCO Post Staff
Gilberto de Lima Lopes, Jr, MD, MBA, of Sylvester Comprehensive Cancer Center at the University of Miami, and Oladimeji Akinboro, MD, MPH, of the U.S. Food and Drug Administration (FDA), discuss a data analysis, which suggests that most subgroups of patients with advanced non–small cell lung cancer with a PD-L1 score of 50% or greater who are receiving FDA-approved chemotherapy/immunotherapy regimens may have overall survival outcomes comparable to or better than immunotherapy-alone regimens (Abstract 9000).
The ASCO Post Staff
Timothy J. Whelan, MD, of McMaster University and Hamilton Health Sciences, discusses findings from the LUMINA study, which found that women aged 55 or older who had grade 1–2 T1N0 luminal A breast cancer following breast-conserving surgery and were treated with endocrine therapy alone had very low rates of local tumor recurrence at 5 years. These patients, the research suggests, may be able to forgo radiotherapy (Abstract LBA501).
The ASCO Post Staff
Carryn M. Anderson, MD, of the University of Iowa Hospital, discusses phase III results of the ROMAN trial of avasopasem manganese for patients with severe oral mucositis who are receiving chemoradiotherapy for locally advanced, nonmetastatic head and neck cancer. Compared with placebo, avasopasem manganese improved severe oral mucositis (Abstract 6005).
The ASCO Post Staff
Thomas Powles, MD, PhD, of Barts Health NHS Trust, Queen Mary University of London, and Jonathan E. Rosenberg, MD, of Memorial Sloan Kettering Cancer Center, discuss the 24-month findings from the phase III EV-301 trial, which suggest that enfortumab vedotin-ejfv continues to show a significant and consistent survival advantage over standard chemotherapy in patients with previously treated advanced urothelial carcinoma (Abstract 4516).
The ASCO Post Staff
Clifford A. Hudis, MD, of the American Society of Clinical Oncology, and Karen E. Knudsen, PhD, MBA, of the American Cancer Society, discuss their collaboration, pooling their research and education resources to help empower patients with cancer and their families. Within 48 hours, Drs. Hudis and Knudsen were able to gear up a rapid response to the crisis in Ukraine, forming a clinical corps of volunteers to post information online in multiple languages, which helped patients navigate their care in the war-torn region. To date, 300 European cancer organizations have joined their efforts.