Sara Zarnegar-Lumley, MD, on AML: Prognostic Effects of IDH Mutations
2020 ASH Annual Meeting & Exposition
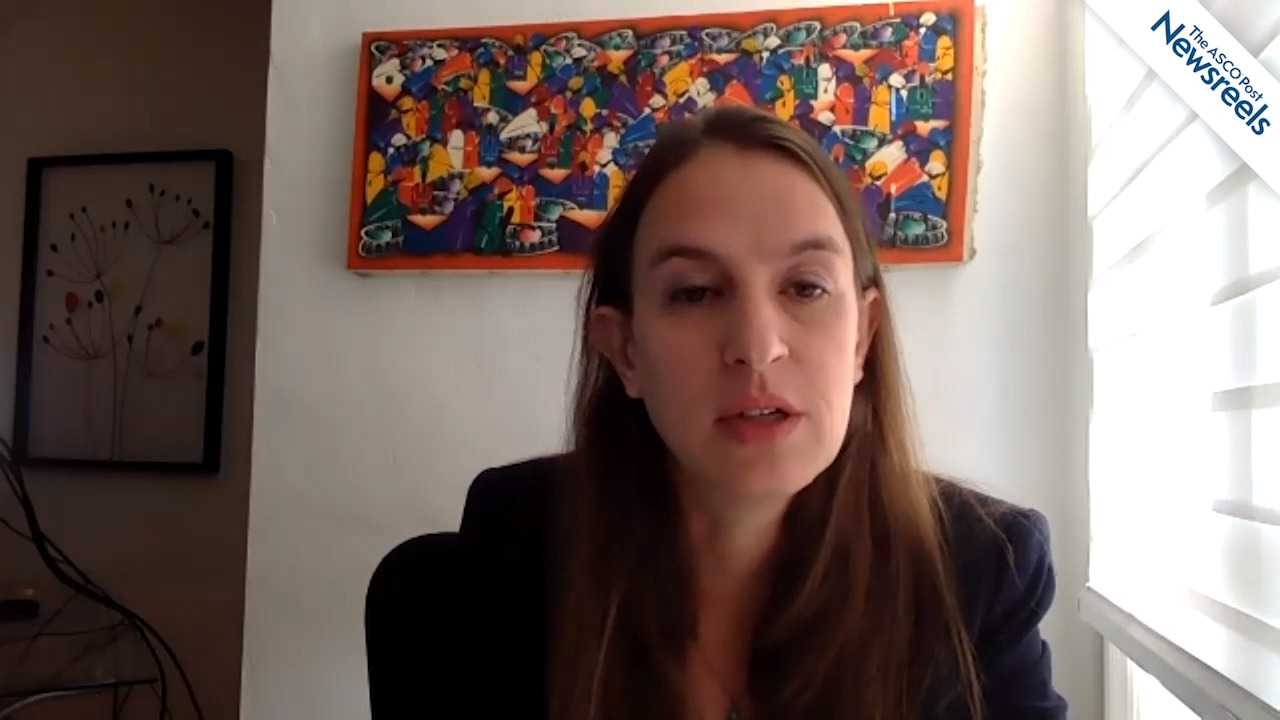
Sara Zarnegar-Lumley, MD, of Vanderbilt University Medical Center, discusses an analysis of a large cohort confirming the age-associated prevalence of IDH mutations in patients, across the age spectrum, with acute myeloid leukemia and therapeutic implications. IDH-mutated genes were found to co-occur frequently with other mutations, some of which favorably impact outcomes in patients younger than 60 (Abstract 388).
The ASCO Post Staff
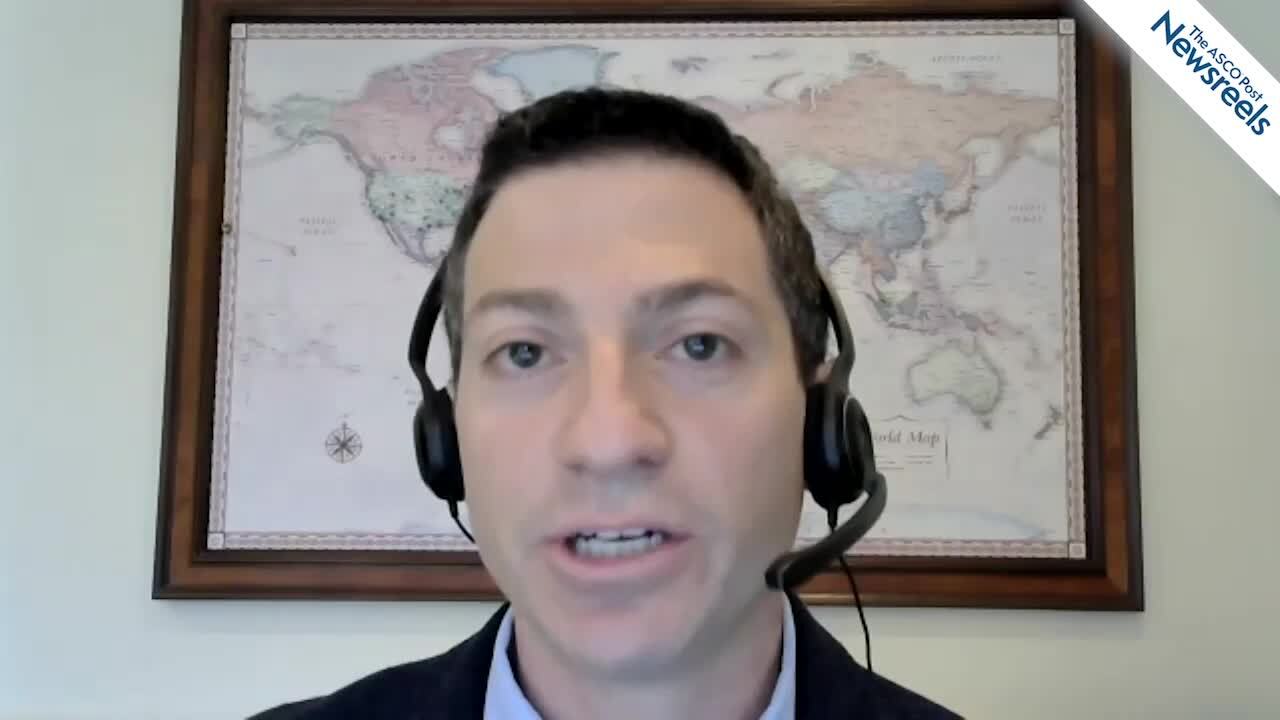
Jorge E. Cortes, MD, of the Georgia Cancer Center at Augusta University, reviews four important studies of treatment advances in chronic myeloid leukemia (CML): nilotinib vs dasatinib in newly diagnosed disease; final 5-year results from the BFORE trial on bosutinib vs imatinib for chronic phase (CP) CML; data from the OPTIC trial on ponatinib for CP-CML; and a novel class of mutated cancer-related genes associated with the Philadelphia translocation (Abstracts 45, 46, 48, 49).
The ASCO Post Staff
Lena E. Winestone, MD, MSHP, of the University of California, San Francisco and Benioff Children’s Hospital, reviews different aspects of bias in treatment delivery, including patient selection for clinical trials; racial and ethnic disparities in survival for indolent non-Hodgkin diffuse large B-cell lymphomas; and end-of-life hospitalization of patients with multiple myeloma, as well as outcome disparities (Abstracts 207-212).
The ASCO Post Staff
Sagar Lonial, MD, of the Emory University School of Medicine, summarizes key papers presented in a session he co-moderated on how second-generation CAR T cells can be used to treat patients with multiple myeloma (Session 653).
The ASCO Post Staff
Matthew S. Davids, MD, of Dana-Farber Cancer Institute, summarizes three key studies from a session he co-moderated on ibrutinib plus venetoclax for first-line treatment of patients with chronic lymphocytic leukemia (CLL) and small lymphocytic lymphoma (SLL), long-term responses to these agents for relapsed and refractory CLL, and undetectable minimal residual disease following fixed-duration treatment with venetoclax and rituximab for CLL (Abstracts 123, 124, and 125).
The ASCO Post Staff
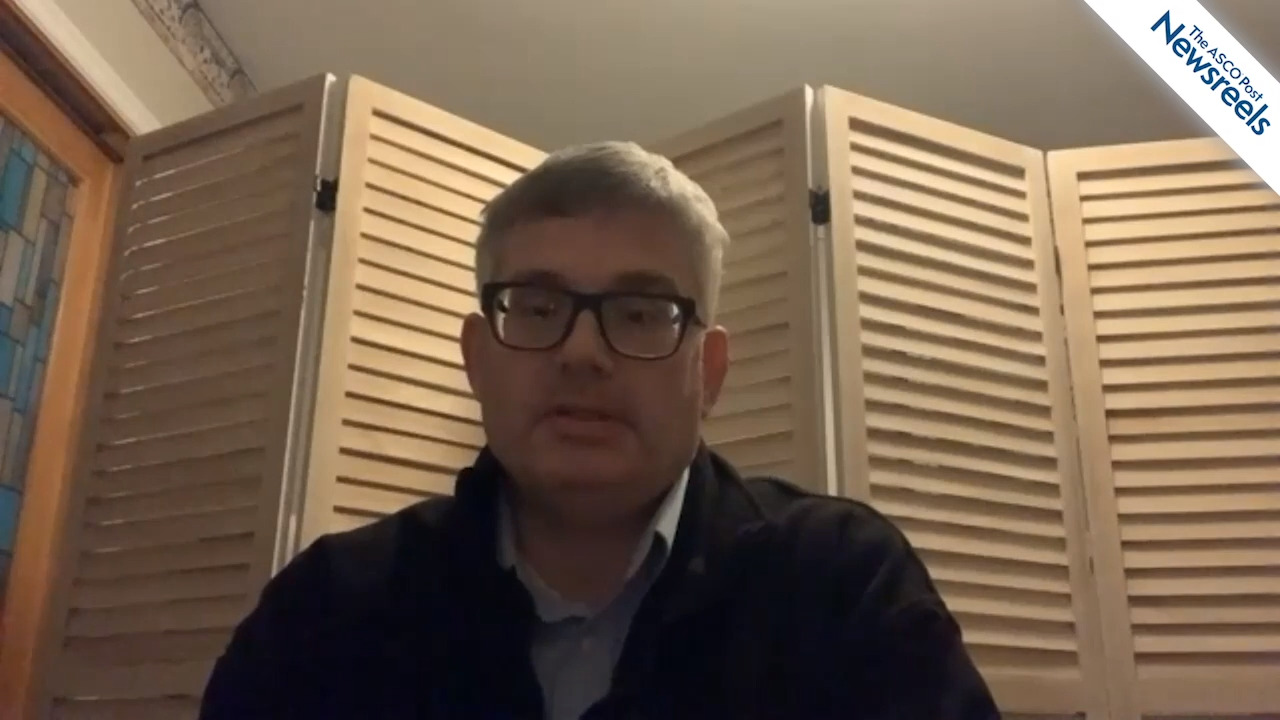
David T. Teachey, MD, of the University of Pennsylvania and Children’s Hospital of Philadelphia, discusses data showing that cranial radiation might be eliminated in most children with T-cell acute lymphoblastic leukemia and that bortezomib may improve survival in children with T-cell lymphoblastic lymphoma (Abstract 266).