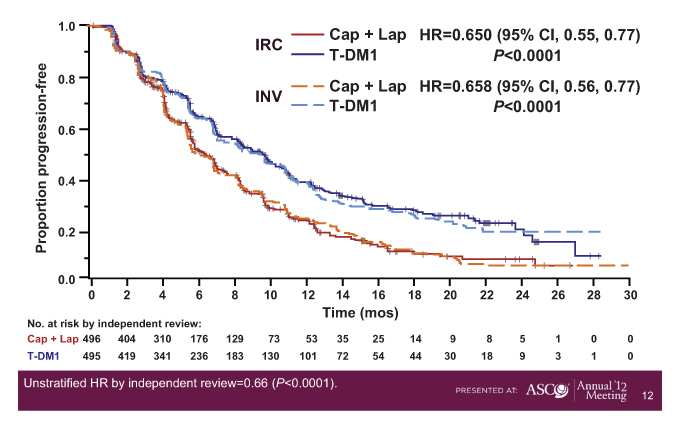
In a study presented at the ASCO Plenary Session, trastuzumab emtansine (T-DM1), the antibody-drug conjugate linking trastuzumab (Herceptin) to a cytotoxic agent, improved progression-free survival by 3.2 months, representing a 35% reduction in risk of progression in the phase III EMILIA trial.1
EMILIA Study Details
EMILIA randomly assigned 991 HER2-positive patients who had locally advanced or metastatic breast cancer and who had previously received a taxane and trastuzumab. Patients received T-DM1 or capecitabine (Xeloda) plus lapatinib (Tykerb) every 3 weeks until progression. Patients experiencing disease progressoin on standard therapy were allowed to cross over to T-DM1.
At the time of this presentation, patients receiving T-DM1 had a disease-free survival of 9.6 months, vs 6.4 months with capecitabine/lapatinib (P < .0001) (Fig. 1).
“T-DM1 readily outpaced capecitabine/lapatininb. All subgroups had relatively comparable benefit,” noted Harold J. Burstein, MD, PhD, of Dana-Farber Cancer Center, Boston, who presented the findings at the Best of ASCO Boston meeting.
Median overall survival was not reached with TDM-1, and was 23.3 months with capecitabine/lapatinib, for a 38% reduction in mortality risk that was not statistically significant. More than 65% of the T-DM1 recipients were alive at the time of analysis, compared to 47.5% of the capecitabine/lapatinib group.
An equally striking difference was in tolerability. Dose reductions were required for only 16.3% of the T-DM1 arm, compared to 53.4% with capecitabine and 27.3% with lapatinib. Patient-reported outcomes were also significantly better with T-DM1.
“This is a remarkably well tolerated product, and that’s exciting,” Dr. Burstein said. “It lacks the typical side effects of chemotherapy because its small dose is mostly delivered selectively to the tumor site.” Because of its strong efficacy and minimal toxicity, “T-DM1 will become a standard second-line treatment in HER2-positive disease,” he predicted.
Editors’ note: New data from the phase III EMILIA study were reported in a press release issued by Genentech on August 26 2012. These updated results showed that T-DM1 significantly improved overall survival in HER2-positive metastatic breast cancer compared to the combination of lapatinib and capecitabine. See the September 15, 2012, issue of The ASCO Post (page 32) for more information, or visit ASCOPost.com.
Trastuzumab Superior to Lapatanib
Meanwhile, the phase III Canadian MA.31/GSK EGF 108919 trial compared current anti-HER2 agents in the first-line metastatic setting and found trastuzumab to be superior to lapatinib, each given with a taxane (weekly paclitaxel or docetaxel every 3 weeks) until progression.2 In the study of 652 patients, median progression-free survival was 8.8 months with lapatinib vs 11.4 months with trastuzumab, indicating a 33% increased risk of progression with lapatinib (P = .01). In women with centrally confirmed HER2-overexpressing tumors, the difference between the arms approached 5 months (P = .003), though overall survival differences have not been observed.
Lapatinib therapy was associated with more adverse events than trastuzumab, most notably diarrhea, and more treatment discontinuations.
Combined HER2 Blockade: Best Neoadjuvant Strategy?
Five large trials have shown that trastuzumab plus lapatinib yields higher pathologic complete response rates than trastuzumab alone, and the most recent was presented at the ASCO Annual Meeting.3 National Surgical Adjuvant Breast and Bowel Project (NSABP) B-41 involved 529 patients who received four cycles of doxorubicin plus cyclophosphamide (AC) followed by weekly paclitaxel (P) plus either trastuzumab, lapatinib, or trastuzumab plus lapatinib prior to surgery.
The pathologic complete response rate was 52.5% in the trastuzumab arm, 53.2% in the lapatinib arm, and 62% with trastuzumab plus lapatinib, for a trend favoring the combination (P = .095). By hormone receptor status, the combination yielded a numeric, though not a statistically significant, benefit over the single agents and was higher among hormone receptor–negative patients (73.0%) than hormone receptor–positive patients (55.6%), which Dr. Burstein called an “exciting” result for HER2-positive, estrogen receptor–negative disease.
When defined as absence of residual disease in both the breast and nodes, pathologic complete responses were achieved by 60.2% with the combination, 49.4% with trastuzumab, and 47.4% with lapatinib, for a strong trend favoring the combination (P = .056).
“In B-41, trastuzumab plus lapatinib was better. Now we have five trials, including two NCI-supported studies, all showing the same result, yet no one is calling the combination an important advantage and no one is moving forward with this regimen,” he said. “Why? That’s a good question with no real answer.” ■
Disclosure: Dr. Burstein reported no potential conflicts of interest.
References
1. Blackwell KL, Miles D, Gianni L, et al: Primary results from EMILIA, a phase 3 study of trastuzumab emtansine (T-DM1) versus capecitabine and lapatinib in HER2-positive locally advanced or metastatic breast cancer previously treated with trastuzumab and a taxane. 2012 ASCO Annual Meeting. Abstract LBA1. Presented June 3, 2012.
2. Gelmon K, Boyle F, Kaufman B, et al: Open-label phase III randomized controlled trial comparing taxane-based chemotherapy with lapatininb or trastuzumab as first-line therapy for women with HER2+ metastatic breast cancer: Interim analysis of NCIC CTG MA.31/GSK EGF 108919. 2012 ASCO Annual Meeting. Abstract LBA671. Presented June 3, 2012.
3. Robidoux A, Tang G, Rastogi P, et al: Evaluation of lapatinib as a component of neoadjuvant therapy for HER2+ operable breast cancer: NSABP protocol B-41. 2012 ASCO Annual Meeting. Abstract LBA506. Presented June 3, 2012.