In patients with advanced or metastatic esophageal squamous cell carcinoma, the addition of the checkpoint inhibitor tislelizumab to first-line chemotherapy significantly improved overall survival vs chemotherapy alone, according to an interim analysis of the global phase III RATIONALE-306 trial. The findings were reported by Harry H. Yoon, MD, Associate Professor of Oncology at the Mayo Clinic Comprehensive Cancer Center, at the European Society for Medical Oncology (ESMO) World Congress on Gastrointestinal Cancers 2022.1

Harry H. Yoon, MD
“The primary endpoint was met, with a statistically significant and clinically meaningful improvement in overall survival,” Dr. Yoon said. “The overall survival benefit with tislelizumab plus chemotherapy was accompanied by significant improvements in progression-free survival and objective response rate, with a more durable tumor response as compared with placebo plus chemotherapy.”
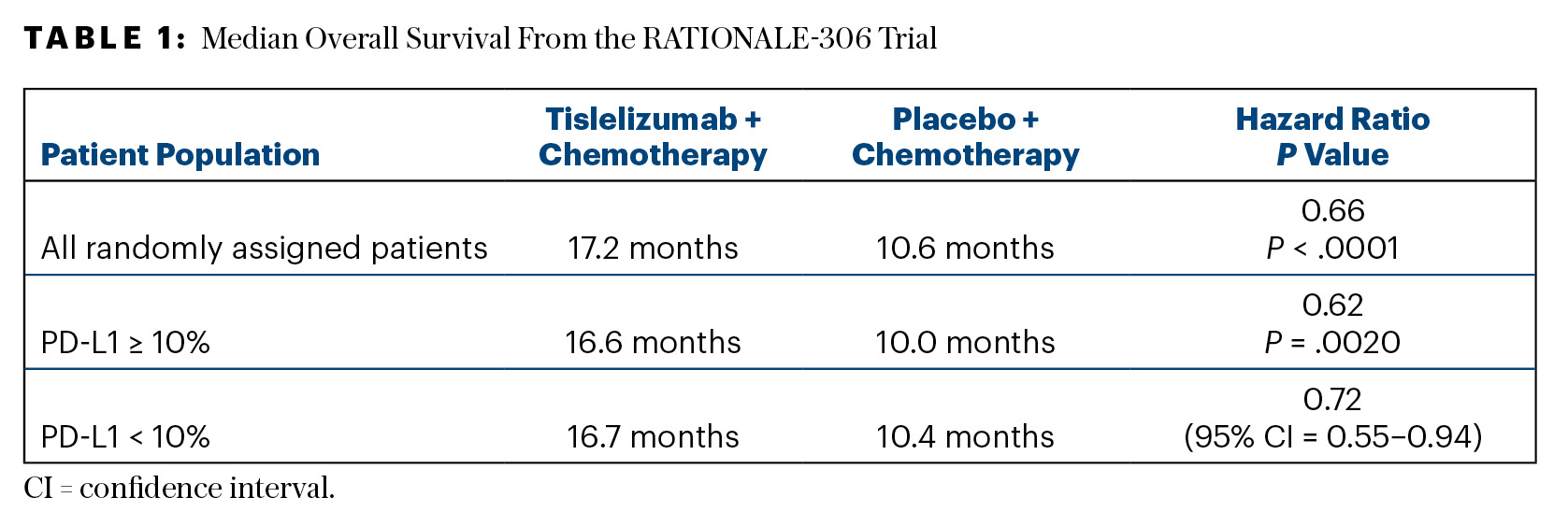
In all randomly assigned patients, median overall survival was 17.2 months vs 10.6 months (hazard ratio [HR] = 0.66; P < .0002). The benefit was consistent across all prespecified subgroups, including according to the expression status of PD-L1.
“The results of the RATIONALE-306 study support tislelizumab plus chemotherapy as a standard first-line treatment option,” Dr. Yoon maintained.
About RATIONALE-306
Tislelizumab, an anti–PD-1 antibody, has high affinity and specificity for PD-1. In RATIONALE-302, the drug yielded a survival benefit as second-line monotherapy in this patient group.2 The addition of anti–PD-1 antibodies to first-line chemotherapy has been shown to improve survival in patients with advanced or metastatic esophageal squamous cell carcinoma, but global phase III trials to date have evaluated these agents only in combination with cisplatin plus fluorouracil.
The global phase III RATIONALE-306 paired the drug with investigator’s choice of chemotherapy, which could be cisplatin or oxaliplatin plus fluoropyrimidine or paclitaxel. The study randomly assigned 649 patients with no prior treatment for unresectable locally advanced or metastatic disease to tislelizumab (200 mg every 3 weeks) or matching placebo plus chemotherapy until disease progression. The primary endpoint was overall survival in all randomly assigned patients.
Among the 649 patients, approximately 86% had metastatic disease, and 37% had a PD-L1 score of at least 10%. More than half the patients received platinum plus paclitaxel, and about half also received additional systemic therapy upon disease progression.
Multiple Endpoints Met
In all randomly assigned patients, median overall survival was 17.2 months with tislelizumab plus chemotherapy and 10.6 months with chemotherapy alone (HR = 0.66; P < .0001). At 12 months, 65.0% and 44.9%, respectively, were alive, as were 48.6% vs 34.5%, respectively, at 18 months. An overall survival benefit was observed regardless of baseline PD-L1 expression status (Table 1).
Median progression-free survival was also significantly improved with tislelizumab plus chemotherapy: 7.3 months vs 5.6 months (HR = 0.62; P < .0001). Tumor response was greater and more durable with the combination, which produced responses in 63.5% vs 42.4% with chemotherapy alone (odds ratio [OR] = 2.38; P < .0001) and complete responses in 4.6% vs 2.5%, respectively. The median duration of response was 7.1 months vs 5.7 months, respectively, and almost twice as many patients had ongoing responses at data cutoff (19.3% vs 9.5%).
Safety Profile
The combination had a manageable safety profile. Adverse events grade ≥ 3 were observed in 66.7% of the combination arm and 64.5% of the chemotherapy-alone arm, leading to death in 1.9% (six patients) and 1.2% (four patients), respectively. Approximately 32% vs 22% of patients discontinued treatment because of toxicity. Approximately one in five patients on tislelizumab plus chemotherapy had an immune-mediated adverse event (almost 9% of which were grade 3).
DISCLOSURE: Dr. Yoon reported financial relationships with ALX Oncology, Novartis, Astellas, Amgen, OncXerna, Merck, Zymeworks, MacroGenics, BMS, BeiGene, and AstraZeneca.
REFERENCES
1. Yoon HH, Kato K, Raymond E, et al: RATIONALE-306: Randomized, global, placebo-controlled, double-blind phase 3 study of tislelizumab plus chemotherapy versus chemotherapy as a first-line treatment for advanced or metastatic esophageal squamous cell carcinoma. ESMO World Congress of Gastrointestinal Cancer 2022. Abstract LBA1. Presented June 29, 2022.


