Longer-term follow-up of the global phase III monarchE trial showed an increasing benefit for adding abemaciclib to endocrine therapy in the adjuvant treatment of early high-risk hormone receptor–positive, HER2-negative breast cancer, regardless of Ki67 index. The latest findings were reported at a European Society for Medical Oncology (ESMO) Plenary Session by Joyce O’Shaughnessy, MD, the Celebrating Women Chair in Breast Cancer Research, Baylor University Medical Center, and Chair of the Breast Cancer Program, Texas Oncology, and the US Oncology Network, Dallas.1 They were also published in Annals of Oncology.2

Joyce O’Shaughnessy, MD

Aditya Bardia, MD, MPH
“The therapeutic regimen of abemaciclib plus endocrine therapy is a major advance in the field of hormone receptor–positive breast cancer,” said the invited discussant of monarchE, Aditya Bardia, MD, MPH, of Massachusetts General Cancer Center, Harvard Medical School, Boston. “The last regulatory approval in the adjuvant hormone receptor–positive setting was more than 15 years ago.”
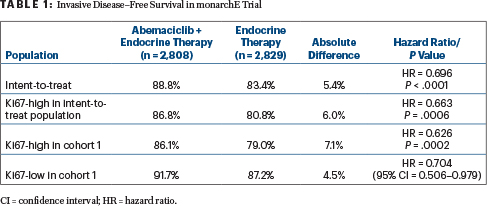
In the new analysis, treatment with abemaciclib plus endocrine therapy reduced the risk of developing an invasive disease–free survival event by 30% and experiencing distant relapse by 31%, at 3 years. The benefit was even greater in patients with a high Ki67 index (Table 1), Dr. O’Shaughnessy reported.
With abemaciclib plus endocrine therapy, vs endocrine therapy alone, invasive disease–free survival rates were 88.8% and 83.4%, respectively (hazard ratio [HR] = 0.696; P < .0001). The distant recurrence–free survival rate was 90.3% vs 86.1%, respectively (hazard ratio [HR] = 0.687; P < .0001). The findings reflect absolute improvements of 5.4% and 4.2%, respectively.
“With further follow-up, the Kaplan-Meier curves have continued to separate, and the treatment benefit was maintained beyond the 2-year treatment period of abemaciclib and endocrine therapy. Given that more than 500 patients are at risk of recurrence at 36 months, we are now able to estimate the 3-year invasive disease–free survival rates, and we showed an absolute difference of 5.4% between the arms,” she reported.
In the previous primary outcome analysis, performed after 19 months of follow-up, invasive disease–free survival was 92.3% with the combination and 89.3% with endocrine therapy alone (HR = 0.71; P = .0009).3
Abemaciclib plus endocrine therapy was recently approved by the U.S. Food and Drug Administration (FDA) for the adjuvant treatment of patients with hormone receptor–positive, HER2-negative, node-positive early breast cancer considered at high risk of recurrence who have a Ki67 index score ≥ 20%. Abemaciclib is the first inhibitor of cyclin-dependent kinases 4 and 6 for adjuvant therapy in breast cancer.
First Analysis, Latest Analysis
At the first interim analysis, monarchE showed a clinically meaningful improvement in invasive disease–free survival and distant relapse–free survival. However, the median follow-up was 15.5 months, and just 12% of patients had completed the 2-year study treatment. The subsequent primary outcome analysis was performed at a median of 19.1 months, with 25% completing study treatment.
The latest “additional follow-up” analysis occurred at 27.1 months, with 89.6% off study, including 72% who completed the 2 years of treatment and 17% who discontinued therapy prematurely. It was performed at the request of regulatory authorities and explored the role of Ki67, a marker of cellular proliferation, as a prognostic and predictive biomarker.
“We’ve seen increasing magnitude of invasive disease–free survival and distant relapse–free survival effect sizes from the first year to the second year, with maintained treatment benefit beyond the 2-year study treatment period,” she said.
A piecewise hazard ratio was estimated using a piecewise exponential model to assess the yearly treatment effect sizes. For invasive disease–free survival, these hazard ratios favoring the combination were 0.795 for year 0 to 1, 0.681 for year 1 to 2, and 0.596 for year 2+. For distant relapse–free survival, they were 0.732, 0.675, and 0.692, respectively.
Ki67 Index Findings
A secondary objective was to test the superiority of abemaciclib plus endocrine therapy vs endocrine therapy alone in patients whose tumors had a high Ki67 index in both the intent-to-treat population as well as in patients enrolled in cohort 1. In patients with a Ki67 index ≥ 20%, benefit was maintained, with invasive disease–free survival events reduced by 37% with abemaciclib/endocrine therapy. The absolute difference between the arms was 7.1% at 3 years.
As expected, patients with a high Ki67 index had worse outcomes, validating Ki67 as a prognostic marker. However, the benefit with abemaciclib was consistent regardless of Ki67 index; the combination reduced the risk by 37% in the Ki67-high population and by 30% in the Ki67-low population.
About monarchE
The global, phase III, monarchE trial randomly assigned 5,637 patients deemed at high risk for recurrence to adjuvant endocrine therapy for at least 5 years, with or without abemaciclib for 2 years. High risk was defined by a compilation of clinical and pathologic factors including nodal status, tumor size, grade, and Ki67 level.
Cohort 1 enrolled patients (56% postmenopausal, 44% premenopausal) with at least four positive axillary lymph nodes or one to three positive nodes and either grade 3 disease or a tumor ≥ 5 cm. Cohort 2 enrolled patients with one to three positive nodes and a centrally determined high Ki67 index (≥ 20%). Some 44.3% of all randomly assigned patients and 39.1% of patients in cohort 1 had tumors with a high Ki67 index. The primary endpoint was invasive disease–free survival in the intent-to-treat population, which comprised both cohorts.
Abemaciclib/Endocrine Therapy: For Which Patients?
During the discussion period, Dr. O’Shaughnessy was asked to whom she would prescribe adjuvant treatment with abemaciclib plus endocrine therapy.
“I think the FDA-approved subset is an appropriate group of patients for us to aim this regimen at for 2 years of adjuvant endocrine therapy. These patients have a very high risk of recurrence. They are refractory to primary endocrine therapy and clearly need additional options,” she responded. In the study, she noted, 16.6% of patients with a Ki67 index ≥ 20% treated with endocrine therapy alone relapsed within 3 years, thus manifesting primary resistance.
No New Safety Signals
Overall, with a median treatment duration of almost 24 months, the adverse-event profile was similar to that in prior analyses. There were “minimal increases” in the development of adverse events of any grade or grade ≥ 3, and the safety profile “remains acceptable for patients with early breast cancer treated with curative intent,” she stated.
Dr. O’Shaughnessy acknowledged that many patients develop some degree of diarrhea; in the study, 6.5% discontinued abemaciclib as a result, but this side effect can be managed with dietary adjustments, loperamide, and dose reductions. “Patients with severe diarrhea need to know to stop the drug,” she said. “It’s generally manageable, but 40% of patients will require a dose adjustment.”
DISCLOSURE: Dr. O’Shaughnessy has received honoraria serving as a consultant or advisor to AbbVie, Agendia, Amgen Biotechnology, AstraZeneca, Bristol Myers Squibb, Celgene, Eisai, Genentech, Genomic Health, GRAIL, Immunomedics, Heron Therapeutics, Ipsen Biopharmaceuticals, Jounce Therapeutics, Eli Lilly, Merck, Myriad, Novartis, O donate Therapeutics, Pfizer, Puma Biotechnology, Prime Oncology, Roche, Seattle Genetics, Syndax Pharmaceuticals, and Takeda.
REFERENCES
1. O’Shaughnessy J, Rastogi P, Harbeck N, et al: Adjuvant abemaciclib combined with endocrine therapy: Updated results from monarchE. ESMO Virtual Plenary Session. Presented October 14, 2021.
2. Harbeck N, Rastogi P, Martin M, et al: Adjuvant abemaciclib combined with endocrine therapy for high-risk early breast cancer: Updated efficacy and Ki67 analysis from the monarchE study. Ann Oncol. September 29, 2021 (early release online).
3. O’Shaughnessy JA, Johnson S, Harbeck N, et al: Primary outcome analysis of invasive disease-free survival for monarchE. 2020 San Antonio Breast Cancer Symposium. Abstract GS1-01. Presented December 9, 2020.

