The antibody-drug conjugate fam-trastuzumab deruxtecan-nxki (T-DXd) may become a new option as a second-line treatment of patients with HER2-positive unresectable or metastatic breast cancer, based on results from the global phase III DESTINY-Breast03 trial. These findings were presented by Javier Cortés, MD, PhD, during a Presidential Symposium of the European Society for Medical Oncology (ESMO) Congress 2021.1
Treatment with T-DXd led to a highly significant 72% reduction in the risk of disease progression vs trastuzumab emtansine (T-DM1) in patients previously treated with a taxane and trastuzumab (hazard ratio [HR] = 0.2840; P = 7.8 × 10–22), according to Dr. Cortés, Director of the International Breast Cancer Center (IBCC), Barcelona and Madrid. The benefit was seen across subgroups, including patients with brain metastases (HR = 0.3796), and for key secondary endpoints. A trend has emerged as well in overall survival, though these data remain immature, he said.

Javier Cortés, MD, PhD

Shanu Modi, MD
“This is terrific news for patients…. In my opinion, based on these data, T-DXd should replace T-DM1 as the standard of care for patients who have previously received trastuzumab and a taxane,” Dr. Cortés said.
Invited study discussant, Shanu Modi, MD, of Memorial Sloan Kettering Cancer Center, New York, agreed: “I believe the DESTINY-Breast03 data support T-DXd as a new second-line option. The efficacy in this randomized trial is unprecedented, and the secondary endpoints and subgroup analyses likewise favor T-DXd…. T-DXd is now my preferred antibody-drug conjugate in the second-line setting.”
Dr. Modi observed: “The progression-free survival curves from DESTINY-Breast03 are absolutely startling. I don’t believe I’ve seen a hazard ratio like this in HER2-positive breast cancer…. Of note, T-DXd delivered in this early-line setting was associated with less lung toxicity [than in DESTINY-Breast01], resetting the risk/benefit analysis in its favor.”
About T-DXd and DESTINY-Breast03
Trastuzumab deruxtecan is an antibody-drug conjugate with three components: a humanized anti-HER2 IgG1 monoclonal antibody with the same amino acid sequence as trastuzumab; a topoisomerase I inhibitor payload; and a tetrapeptide-based cleavable linker. The drug was approved by the US Food and Drug Administration in December 2019 for the treatment of patients with unresectable or metastatic HER2-positive breast cancer who have received at least two prior anti–HER2-based regimens, based on the phase II DESTINY-Breast01 trial.2
The subsequent open-label randomized DESTINY-Breast03 trial is the first global phase III head-to-head trial of T-DXd against an active control in patients with HER2-positive metastatic breast cancer following initial treatment with trastuzumab and a taxane. The study randomly assigned 524 previously treated patients from North America, South America, Europe, Asia, and Oceania to T-DXd at 5.4 mg/kg or T-DM1 at 3.6 mg/kg every 3 weeks. The primary endpoint was progression-free survival by blinded independent central review.
Primary Endpoint Met
At a median follow-up of about 16 months, median progression-free survival by blinded independent central review was not reached with T-DXd and was 6.8 months with T-DM1, a risk reduction of 74% (P = 7.8 × 10–22). The estimated 12-month overall survival rate was 94.1% vs 85.9% (HR = 0.5546; P = .007172), which did not cross the prespecified boundary for significance. Other endpoints were similarly improved (Table 1).
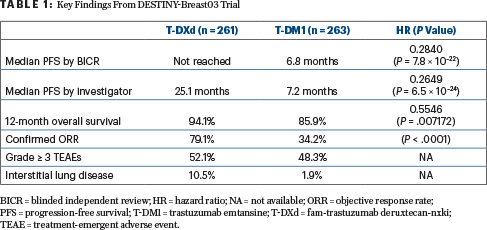
Safety Profile
Safety results from the study showed consistency with prior clinical trials. Similar rates of treatment-emergent adverse events were observed between the arms, and no new safety signals emerged.
Of special note, there was an improvement in the interstitial lung disease profile as compared with studies performed in more heavily pretreated patients. Adjudicated drug-related interstitial lung disease occurred in 10.5% of the T-DXd arm, most of which (9.7%) were grade 1 or 2, and none were grade 4 or 5. This toxicity was observed in 1.9% of the T-DM1 arm, and all were grade 1 or 2. Decreases in left-ventricular ejection fraction were seen in 2.7% of the T-DXd arm, all grade 1 or 2, and in 0.4% of the T-DM1 arm (grade 2). More than one-third of patients on the T-DXd arm experienced alopecia, and for one patient, it was grade ≥ 3.
Drug-related adverse events associated with dose reductions were significantly more common with T-DXd (21.4% vs 12.6%), as were those associated with treatment discontinuation (12.8% vs 5.0%).
Looking Backward, Looking Ahead
DESTINY-Breast03 moves T-DXd forward and builds strongly upon DESTINY-Breast01. In the older study, single-agent T-DXd yielded a response rate of 60.3% and a disease control rate was 97.3%; median progression-free survival was 16.4 months (18.1 months among 24 patients with brain metastases), and median overall survival was not reached.
Dr. Cortés concluded: “We now need to understand whether we can have the same great data in the first-line setting and, hopefully, even in early breast cancer. Trials are ongoing.”
Future Trials With T-DXd or T-DM1 in HER2-Positive Breast Cancer
First-Line Trials
DESTINY-Breast09: T-DXd vs T-DXd plus pertuzumab vs taxane plus trastuzumab and pertuzumab (N = 1,134)
NSABP B-60/DESTINY-Breast05: T-DM1 vs T-DXd in high-risk patients (N = 1,600)
Second-Line Trial
HER2CLIMB-02: tucatinib plus T-DM1 vs T-DM1 (N = 460)
ADJUVANT THERAPY Trial
ATEMPT 2.0: T-DM1 followed by trastuzumab to paclitaxel and trastuzumab (N = 501) in patients with stage I disease
RESIDUAL DISEASE Trial
CompassHER2 RD: T-DM1 vs T-DM1 plus tucatinib in patients with residual disease after neoadjuvant therapy (N = 1,031)
Trial IN CNS METASTASES
TUXEDO-1 phase II: T-DXd in patients with newly diagnosed or progressive brain metastases (N = 15)
CNS = central nervous system; T-DM1 = trastuzumab emtansine;
T-DXd = fam-trastuzumab deruxtecan-nxki.
DISCLOSURE: Dr. Cortes has served on advisory boards or on speakers bureaus or has received travel funding from Roche, Celgene, Cellestia, AstraZeneca, Biothera, Merus, Seattle Genetics, Daiichi Sankyo, Erytech, Athenex, Polyphor, Lilly, Servier, Merck Sharp & Dohme, GlaxoSmithKline, Leuko Bioscience, Bioasis, Clovis Oncology, Boehringer, Kyowa Kyrin, 4D Pharma PLC, Novartis, Eisai, Pfizer, and Samsung Bioepis; holds stock in MedSIR; and has received research grants from numerous companies. Dr. Modi has reported financial relationships (research, consultancy, and advisory) with Novartis, AstraZeneca/MedImmune, Seattle Genetics, Daiichi Sankyo, Carrick, Eli Lilly, MacroGenics, and GSK.
REFERENCES
1. Cortés J, Kim S, Chung W, et al: Trastuzumab deruxtecan (T-DXd) vs trastuzumab emtansine (T-DM1) in patients with HER2+ metastatic breast cancer: Results of the randomized phase III DESTINY-Breast03 study. ESMO Congress 2021. Abstract LBA1. Presented September 18, 2021.
2. Krop IE, Saura C, Yamashita T, et al: [Fam-] trastuzumab deruxtecan (T-DXd; DS-8201a) in subjects with HER2-positive metastatic breast cancer previously treated with T-DM1: A phase 2, multicenter, open-label study (DESTINY-Breast01). 2019 San Antonio Breast Cancer Symposium. Abstract GS1-03. Presented December 11, 2019.

