
Hagop Kantarjian, MD, FASCO

Emil J. Freireich, MD, FASCO
With the accelerating pace of research progress in leukemia comes the challenge of timely translation of discoveries into clinical practice. This is particularly relevant in subtypes characterized by many exciting therapeutic advances.
These disease entities include Philadelphia chromosome (Ph)-positive and B-cell acute lymphoblastic leukemia (ALL), with the development of novel, more potent, and safer BCR::ABL1 tyrosine kinase inhibitors (TKIs), as well as CD19/20/22-targeting antibody-drug conjugates (eg, inotuzumab ozogamicin [anti-CD22]), bispecific T-cell engagers (eg, blinatumomab [anti-CD19]), and engineered chimeric antigen receptor (CAR) T-cell therapies. Another example is chronic lymphocytic leukemia (CLL), with the availability of more potent and safer Bruton’s tyrosine kinase (BTK) inhibitors (eg, acalabrutinib, zanubrutinib, pirtobrutinib) and degraders, the BCL2 inhibitor venetoclax and subsequent novel compounds, and recent data that 2 years of combination therapy with a BTK inhibitor and venetoclax may produce deep molecular remissions that can last for the patient’s lifetime.1-4
In such a rich research environment, relying on large-scale randomized trials in rare cancers such as leukemias may slow progress, because when these trials take 5 to 10 years to mature, their results may become less relevant to emerging therapeutic realities. Clinical research may need to shift to well-designed Bayesian single-arm trials, incorporating comparisons with historical data, real-world data, and synthetic controls, until an optimal regimen is considered ready for comparison with an acceptable standard-of-care control arm.
For simplicity, this overview divides the leukemias into favorable, intermediate, and unfavorable categories based on estimated 5- to 10-year overall survival rates. In 1980, acute promyelocytic leukemia (APL) was the sole entity in the intermediate category, with a cure rate of approximately 40%; all other leukemias were unfavorable. Chronic myeloid leukemia (CML) and CLL were incurable, and the cure rates of acute myeloid leukemia (AML) and adult ALL with chemotherapy were approximately 20% to 30%.1-4
Today, most leukemias have shifted into the favorable category (defined as those with current 5-year overall survival rates > 70%), including hairy cell leukemia (HCL), APL, core-binding factor AML, Ph-positive CML, CLL, and, most recently, adult Ph-positive ALL and B-cell ALL in younger patients (aged < 60 years). Intermediate leukemias have current 5-year overall survival rates of approximately 40% to 70%, represented by older patients with B-cell ALL (aged ≥ 60 years) and younger patients with AML (aged < 60 years). Unfavorable leukemias have 5-year overall survival rates below 40% and consist of older patients with AML (aged ≥ 60 years), treated secondary AML (eg, arising from myelodysplastic syndrome or myeloproliferative neoplasm), AML with complex karyotype, TP53-mutated AML, MECOM-rearranged AML, and other rare subsets.1-4
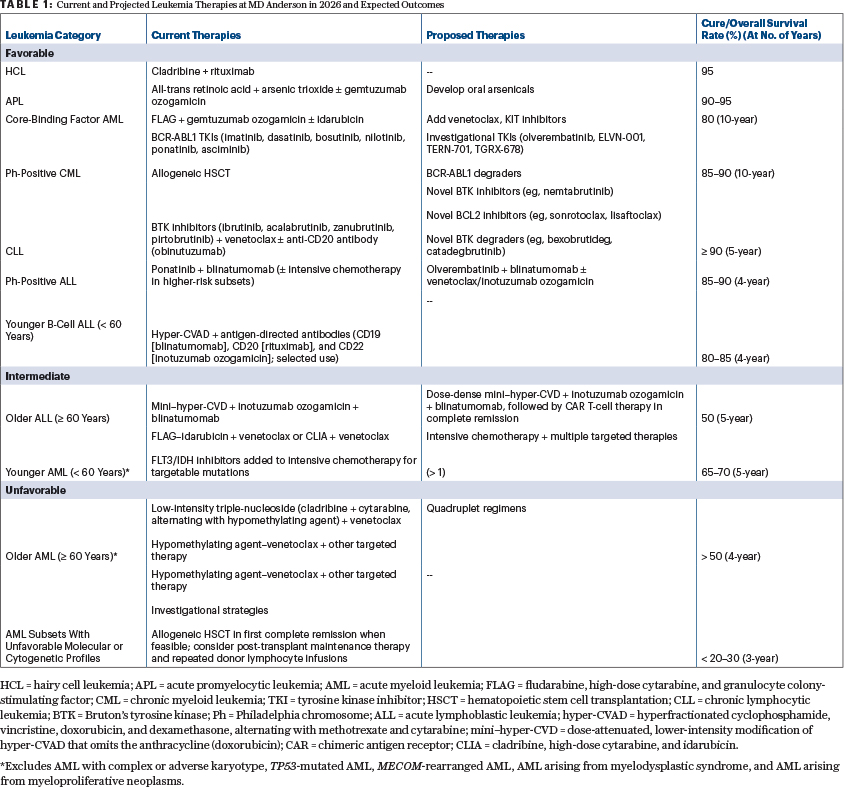
This brief review in The ASCO Post offers practicing oncologists a simplified high-level overview and resources for additional information about the current research progress in leukemia, as summarized in Table 1.1-4
The Favorable Leukemias
Hairy Cell Leukemia (HCL)
Cladribine, alongside other adenosine nucleoside analogs (ie, pentostatin), is used in the treatment of HCL and is highly effective, as is the anti-CD20 monoclonal antibody rituximab.5,6 The sequential combination of cladribine (5.6 mg/m2 intravenously [IV] daily for 5 days) followed by rituximab (375 mg/m2 IV weekly for 8 weeks, starting 4 weeks later) resulted in a 10-year event-free survival rate of95% in newly diagnosed patients.7
Because BRAF V600E mutations are causal in HCL, studies of BRAF inhibitors (eg, vemurafenib) plus anti-CD20 antibodies (eg, rituximab, obinutuzumab) were conducted and showed promise.8
Acute Promyelocytic Leukemia (APL)
The discovery of the anti-APL efficacy of all-trans retinoic acid and arsenic trioxide led to their combination in a chemotherapy-free regimen at The University of Texas MD Anderson Cancer Center in 2001. In front-line APL, all-trans retinoic acid and arsenic trioxide–based treatment has since produced a complete remission rate of approximately 94% and a 10-year overall survival rate exceeding 90%.9 These results were supported by randomized trials and real-world studies.10-13 Oral arsenic trioxide formulations currently in development will make APL treatment more convenient.14,15
Monitoring for PML-RARA molecular disease in complete remission is critical to identify the rare cases of relapse and to quickly reinitiate therapy that can still be curative. Relapse is curable with longer courses of all-trans retinoic acid and arsenic trioxide, with or without gemtuzumab ozogamicin and/or anthracyclines (if not used in front-line therapy), and with or without autologous or allogeneic hematopoietic stem cell transplantation (HSCT).16
Core-Binding Factor Acute Myeloid Leukemia (AML)
Core-binding factor AML, representing approximately 15% of adult AML and 25% to 30% of pediatric AML cases, includes leukemias characterized by inv(16)(p13.1q22)/t(16;16)(p13.1;q22) and t(8;21)(q22;q22). The addition of four courses of high-dose cytarabine consolidation therapy improved the 5-year overall survival rate from approximately 35% to 50%; adding gemtuzumab ozogamicin increased it to 75%.17,18 Based on data from MD Anderson, the addition of gemtuzumab ozogamicin to fludarabine, high-dose cytarabine, and granulocyte colony-stimulating factor (FLAG; resulting in FLAG-GO) showed a 5-year overall survival rate of approximately 80%.19
Persistent measurable residual disease (MRD) positivity at the end of therapy indicates the need to consider additional interventions (autologous or allogeneic HSCT; hypomethylating agents–venetoclax with or without gemtuzumab ozogamicin).20
Ph-Positive Chronic Myeloid Leukemia (CML)
The development of BCR::ABL1 TKIs changed CML from an invariably fatal disorder before 2000 (in the absence of HSCT) to an indolent, functionally curable disease.21 Patients who are adherent to daily TKI therapy have a near-normal life expectancy, with 10-year relative survival reported at 92%.22-29
Six BCR-ABL1 TKIs are currently commercially available. Therapy with any of the front-line–approved TKIs (first-generation: imatinib; second-generation: dasatinib, bosutinib, nilotinib; third-generation: asciminib [Selectively targets the ABL1 myristoyl pocket, ie, “STAMP” inhibitor]) normalizes survival. None of the newer-generation TKIs have been shown to improve overall survival compared with imatinib, nor to improve long-term rates of treatment-free remission (defined as discontinuation of TKI therapy without subsequent CML recurrence). Treatment-free remission status is essentially a “molecular cure.” In my view, with optimal TKI therapy (discontinuation after ≥ 5 years of durable, sustained deep molecular response [BCR::ABL1 transcripts on the International Scale ≤ 0.01%; MR4+]), the molecular cure rate is at least 30% to 40% (50%–60% of patients ultimately achieve durable deep molecular response; 60%–80% of those with durable deep molecular responses > 5 years achieve treatment-free remission).22-29
All five front-line–approved TKIs, plus ponatinib (third-generation), are used in later-line therapy with excellent efficacy. The third-generation TKIs, so called because they were developed after the second-generation agents, are more potent and can overcome resistance due to an emerging T315I clone (unlike imatinib and the second-generation TKIs). In addition, there are at least four promising third-generation TKIs in clinical trials: olverembatinib and ELVN-001 (like ponatinib, both target the ABL1 kinase domain), and TERN-701 and TGRX-678 (STAMP inhibitors).22-29
To recap, the aims of therapy in CML are:
- To achieve nearly normal survival: The current 10-year relative survival rate is high and unlikely to be substantially improved.
- To increase the rate of treatment-free remission or molecular cure: This may be possible, but no strategy has succeeded so far. Perhaps adding the novel BCR-ABL1 degraders to BCR::ABL1 inhibitors will achieve this goal.
- To help patients maintain a normal quality of life by reducing TKI side effects: This can be achieved thanks to the range of TKIs with differing side effect profiles, along with dose reductions that preserve efficacy (often referred to as “optimal biologic doses”).
- To obtain TKIs at a good “treatment value”: This is also achievable with the availability of generic formulations (eg, imatinib: ~ $400–$500/year; dasatinib [50 mg/day] through CostPlus: < $1,500/year).22-29
Allogeneic HSCT is highly curative but is associated with some mortality (long-term mortality: ~ 10%–15%) and potentially serious toxicities.2 It should be considered in patients with chronic-phase CML who develop resistance to a second-generation TKI without an identifiable guiding mutation, as well as in those with de novo blast phase or subsequent transformation to accelerated or blast phase.
Chronic Lymphocytic Leukemia (CLL)
Today, CLL is considered incurable by many experts. However, if “cure” is defined as achieving a deep molecular response (by next-generation sequencing [NGS]–MRD) without recurrence of CLL requiring subsequent therapeutic intervention in the patient’s lifetime, then it may be attainable today with combinations of BTK inhibitors and venetoclax (given for ≥ 2 years), or with triplet therapy comprising a BTK inhibitor, venetoclax, and an anti-CD20 antibody.
Historically, the median overall survival in CLL was 4 to 5 years, and less than 2 to 3 years once therapy was required. The FCR regimen (fludarabine, cyclophosphamide, and rituximab) demonstrated for the first time the potential curability of CLL, particularly in patients with favorable-risk disease (disease-free survival rate of 40% in those with mutated IGHV). However, FCR use faded rapidly with the advent of oral targeted therapies (BTK inhibitors and venetoclax).30-34
In a study from MD Anderson of patients with treatment-naive CLL receiving 420 mg of ibrutinib daily, with venetoclax added 3 months into therapy and continued for a total of 2 years of combination treatment, the 5-year progression-free survival rate was 90%, and the overall survival rate was 96%.35-37 A more recent trial with a total treatment duration of 13 cycles using pirtobrutinib, venetoclax, and obinutuzumab resulted in 1-year bone marrow MRD–negative rates by NGS of 81% at 10-6 sensitivity and 96% at 10-4 sensitivity, which were higher than with ibrutinib plus venetoclax by flow cytometry at 10-4 sensitivity.35-38 No patients experienced disease progression.38 Other studies investigating ibrutinib and venetoclax combinations, with some modifications, reported similar findings but a higher rate of CLL relapse, perhaps due to shorter treatment duration.34 Two recent randomized trials may lead to pirtobrutinib being approved for use in the front-line setting (in addition to ibrutinib, acalabrutinib, and zanubrutinib).
On the horizon are novel BTK inhibitors (eg, nemtabrutinib), novel BCL2 inhibitors (eg, sonrotoclax, lisaftoclax), and novel BTK degraders (eg, bexobrutideg, catadegbrutinib), the latter of which may eventually replace BTK inhibitors. The most important aim of ongoing CLL research is to optimize treatment modalities in combinations that achieve the highest rates of MRD negativity.
Ph-Positive Acute Lymphoblastic Leukemia (ALL)
Before 2000, adult Ph-positive ALL was almost always fatal (5-year overall survival rate < 10%) without allogeneic HSCT in first complete remission (~ 35%). The two therapeutic inflection points were the addition of BCR::ABL1 TKIs to chemotherapy starting in 2000 and the replacement of intensive chemotherapy with blinatumomab in 2017. This transformed the disease from the worst adult ALL subset to the most favorable.
At MD Anderson, in 2017, we initiated a trial of ponatinib plus blinatumomab in newly diagnosed Ph-positive ALL, in which blinatumomab was given simultaneously with ponatinib during induction and later for a total of five courses. Among the first 84 patients treated, the NGS–MRD negativity rate was 95%, and the 4-year overall survival rate was 88%.2,39 An Italian study that randomly assigned patients to this combination vs imatinib plus chemotherapy showed, as expected, better outcomes with ponatinib plus blinatumomab: a higher MRD response rate at day 133 (after two blinatumomab cycles or four or six chemotherapy cycles; 70% vs 53%; P = .009); and a higher 18-month event-free survival rate (90% vs 77%; P = .011).40
Younger B-Cell ALL
In adult ALL, traditional intensive chemotherapy used over the past 6 decades has resulted in 5-year overall survival rates of 40% to 60% in younger patients (aged < 60 years) and 10% to 20% in older patients. What revolutionized treatment is the discovery of the high efficacy of blinatumomab, inotuzumab ozogamicin, and CAR T-cell therapy.41-44 In 2010 at MD Anderson, following these discoveries, we began developing regimens that combine less chemotherapy with blinatumomab and/or inotuzumab ozogamicin. With hyper-CVAD (hyperfractionated cyclophosphamide, vincristine, doxorubicin, and dexamethasone, alternating with methotrexate and cytarabine) plus sequential blinatumomab with or without fractionated low-dose inotuzumab ozogamicin given to 100 younger patients with newly diagnosed B-cell ALL (updated cohort including the previously published first 75 patients), the complete remission rate was 100%, the NGS–MRD negativity rate was 76%, the 5-year overall survival rate was 87%, and the event-free survival rate was 85%.45
Future modifications may include adding more potent CD20-targeted bispecific T-cell engagers, using the subcutaneous formulation of blinatumomab, incorporating blinatumomab into chemotherapy (dose-dense, starting on days 4–5 for 18 days) rather than using it sequentially, using CAR T-cells in first complete remission to replace allogeneic HSCT, and using NGS–MRD to assess the benefit of therapy and guide treatment changes.44,46,47
The Intermediate Leukemias
Older B-Cell Acute Lymphoblastic Leukemia (ALL)
With intensive chemotherapy, outcomes in older patients with ALL are poor: complete remission rates of 60% to 70%; high mortality in complete remission; and 5-year overall survival rates of 8% to 15%.48 In 2010, MD Anderson studies added inotuzumab ozogamicin to mini–hyper-CVD (a dose-attenuated, lower-intensity modification of hyper-CVAD that omits the anthracycline [doxorubicin]) chemotherapy and later introduced sequential blinatumomab. Among patients with a median age 68 years, the complete remission rate was 89%, and the 5-year overall survival rate was 46% (vs 20%–25% historically in MD Anderson data).49 Several international studies reported similar results when adding inotuzumab ozogamicin or blinatumomab to less-intensive chemotherapy.50,51
Future research will focus on combining blinatumomab and inotuzumab ozogamicin with perhaps less chemotherapy and dose-dense chemotherapy and using CAR T-cell consolidation therapy to improve tolerance, reduce mortality in complete remission, and increase cure.
Younger/Fit Acute Myeloid Leukemia (AML)
There are exhaustive discussions about which patients can tolerate intensive chemotherapy across the age spectrum. The reality, extrapolated from Surveillance, Epidemiology, and End Results (SEER) data in nearly 38,000 patients (1975–2022), is that, in community practice, induction mortality is approximately 33% overall, 15% in patients younger than age 60, 32% in patients aged between 60 and 75 years, and 53% in patients older than age 75.52 Thus, at MD Anderson, we categorize patients as either younger than age 60 and offer them intensive chemotherapy (unless they are in poor condition [eg, poor performance status, pneumonia or pulmonary infiltrates, organ dysfunction]), or older than age 60 and offer them lower-intensity therapies.
The 7 + 3 (cytarabine continuously for 7 days, along with short infusions of an anthracycline on each of the first 3 days) regimen is a poor standard of care that has survived 6 decades without challenge. It results in an approximate 5-year overall survival rate of 35% to 40% in younger patients and 15% at 3 years in older patients who are fit for intensive chemotherapy.53,54

The FLAG–idarubicin (FLAG-IDA) and CLIA (cladribine, high-dose cytarabine, and idarubicin) regimens have shown superior outcomes, which can be further improved with the addition of venetoclax and other targeted therapies, such as FLT3 and IDH1/2 inhibitors. With FLAG-IDA–venetoclax and CLIA–venetoclax, the composite complete remission rates were 95% and 93% (in a predominantly AML cohort), respectively, and 3-year overall survival rates were up to approximately 70%.55,56 Allogeneic HSCT in first complete remission is generally associated with improved outcomes. Combinations of 7 + 3 or 5 + 2 (a truncated 7 + 3 regimen with 5 days of cytarabine and 2 days of an anthracycline) with venetoclax, studied in China and Australia respectively, have also shown favorable results.57,58
Adding FLT3 inhibitors to intensive chemotherapy in FLT3-mutated AML improved outcomes,59 as did non–FLT3-targeting strategies (high-dose cytarabine; high-dose daunorubicin or idarubicin; the addition of gemtuzumab ozogamicin, cladribine, or venetoclax).60,61 In patients with FLT3–wild-type AML but FLT3-like biology (eg, mutations in NPM1, DNMT3A, or FLT3 tyrosine kinase domain), the addition of quizartinib to intensive chemotherapy prolonged overall survival.62 The addition of IDH inhibitors to intensive chemotherapy in IDH1/2-mutated AML was associated with good outcomes, with overall response rates of 74% to 77% and 3-year overall survival rates of 61% to 78%.63 Ongoing trials are combining chemotherapy with menin inhibitors in KMT2A-rearranged AML.
The Unfavorable Leukemias
Older/Unfit Acute Myeloid Leukemia (AML) and Subsets With Unfavorable Molecular or Cytogenetic Profiles
Older adults with AML have historically faced a poor prognosis. Recent studies combining hypomethylating agents with venetoclax in patients unfit for intensive chemotherapy have reported approximately 25% 3-year overall survival.64 Thus, the current accepted standard of care in older or unfit AML remains suboptimal. Early reports of triplet therapies (hypomethylating agent–venetoclax plus a targeted therapy) show encouraging outcomes for these patients.65 Other intensified venetoclax-containing regimens have also been explored; at MD Anderson, pilot studies using a triple-nucleoside regimen (cladribine plus low-dose cytarabine, alternating with a hypomethylating agent) combined with venetoclax have demonstrated excellent results.
Among older or unfit patients treated with the triple-nucleoside–venetoclax regimen, the MRD-negative rate was 84%, the induction mortality rate was 2% (compared with population-based SEER data mortality of ~ 30%–50%), and the 4-year overall survival rate was 52% (compared with a 3-year rate of approximately 25% with hypomethylating agents plus venetoclax).66,67
In newly diagnosed, intensive chemotherapy–ineligible patients with IDH-mutated AML treated with a triplet regimen of a hypomethylating agent, venetoclax, and an IDH inhibitor, the composite complete remission rate was 92%, and the 2-year overall survival rate was 69%.68 Triplet therapy with a hypomethylating agent, venetoclax, and an FLT3 inhibitor resulted in a complete remission rate of 90% and 2-year overall survival rate exceeding 70% in FLT3-mutated AML.69
Regimens incorporating triplet combinations of hypomethylating agents, venetoclax, and menin inhibitors (eg, revumenib, ziftomenib, bleximenib, enzomenib) are currently being studied in KMT2A-rearranged and NPM1-mutated AML.70,71 Triplet regimens may be associated with significant myelosuppression causing mortality, thus requiring careful development of safe schedules.
Summary
In 2014, an ASCO 50th anniversary article reviewed advances in leukemia over the preceding half century.72 The editorial included brief mentions of blinatumomab and inotuzumab ozogamicin in ALL, as well as ibrutinib in CLL. Now, 12 years later, the curative picture in leukemia is much brighter. All 14 drug approvals in AML have occurred since 2017, and the outlook for this and other leukemias has significantly improved.61 Today, leukemia research is taking place in a rich landscape of basic, translational, and clinical discoveries.
For a more detailed review of progress in leukemia, please see References 1 and 2.
DISCLOSURE: This study was funded in part by the National Cancer Institute/National Institutes of Health (NCI/NIH) through the Support Grant P30 CA016672 to The University of Texas MD Anderson Cancer Center and was supported by the Tanoto Foundation and the Charif Souki Cancer Research Fund. Dr. Kantarjian has received research grants and honoraria from AbbVie/ImmunoGen, Amgen, Ascentage, Jazz, KAHR, Novartis, Pfizer, and Stemline.
REFERENCES
- Kantarjian H, Chifotides HT, Haddad FG, et al: The care and cure of the leukemias in 2026. Am J Hematol 101:807-831, 2026.
- Kantarjian HM, Borthakur G, Daver N, et al: A tour of leukemia progress in 2025, viewed through the MD Anderson leukemia research lens. Cancer 131:e70113, 2025.
- Kantarjian HM, Jain N, Garcia-Manero G, et al: The cure of leukemia through the optimist's prism. Cancer 128:240‑259, 2022.
- Kantarjian HM, Keating MJ, Freireich EJ: Toward the potential cure of leukemias in the next decade. Cancer 124:4301‑4313, 2018.
- Rosenberg JD, Burian C, Waalen J, et al: Clinical characteristics and long-term outcome of young hairy cell leukemia patients treated with cladribine: A single-institution series. Blood 123:177‑183, 2014.
- Thomas DA, O’Brien S, Bueso-Ramos C, et al: Rituximab in relapsed or refractory hairy cell leukemia. Blood 102:3906‑3911, 2003.
- Marvin-Peek J, Jen W-Y, Kantarjian HM, et al: Long-term results of the sequential combination of cladribine and rituximab in hairy cell leukemia. Leuk Lymphoma 65:1325‑1334, 2024.
- Tiacci E, De Carolis L, Simonetti E, et al: Vemurafenib plus rituximab in refractory or relapsed hairy-cell leukemia. N Engl J Med 384:1810‑1823, 2021.
- Jen W-Y, Marvin-Peek J, Kantarjian HM, et al: Long-term follow-up of a phase 2 study of all-trans retinoic acid, arsenic trioxide, and gemtuzumab ozogamicin in acute promyelocytic leukemia. Cancer 131:e35662, 2025.
- Lo-Coco F, Avvisati G, Vignetti M, et al: Retinoic acid and arsenic trioxide for acute promyelocytic leukemia. N Engl J Med 369:111‑121, 2013.
- Platzbecker U, Avvisati G, Cicconi L, et al: Improved outcomes with retinoic acid and arsenic trioxide compared with retinoic acid and chemotherapy in non–high-risk acute promyelocytic leukemia: Final results of the randomized Italian-German APL0406 trial. J Clin Oncol 35:605‑612, 2017.
- Burnett AK, Russell NH, Hills RK, et al: Arsenic trioxide and all-trans retinoic acid treatment for acute promyelocytic leukaemia in all risk groups (AML17): Results of a randomised, controlled, phase 3 trial. Lancet Oncol 16:1295‑1305, 2015.
- Voso MT, Guarnera L, Lehmann S, et al: Acute promyelocytic leukemia: Long-term outcomes from the HARMONY project. Blood 145:234‑243, 2025.
- Au W-Y, Kumana CR, Lee HKK, et al: Oral arsenic trioxide–based maintenance regimens for first complete remission of acute promyelocytic leukemia: A 10-year follow-up study. Blood 118:6535‑6543, 2011.
- Ravandi F, Rangaraju S, Kantarjian H, et al: A pharmacokinetic and safety study of oral arsenic trioxide in patients with acute promyelocytic leukemia. Blood Adv 9:2136‑2143, 2025.
- Sasaki K, Ravandi F, Kadia T, et al: Outcome of patients with relapsed acute promyelocytic leukemia. Clin Lymphoma Myeloma Leuk 24:375‑381, 2024.
- Byrd JC, Ruppert AS, Mrózek K, et al: Repetitive cycles of high-dose cytarabine benefit patients with acute myeloid leukemia and inv(16)(p13q22) or t(16;16)(p13;q22): Results from CALGB 8461. J Clin Oncol 22:1087‑1094, 2004.
- Hills RK, Castaigne S, Appelbaum FR, et al: Addition of gemtuzumab ozogamicin to induction chemotherapy in adult patients with acute myeloid leukaemia: A meta-analysis of individual patient data from randomised controlled trials. Lancet Oncol 15:986‑996, 2014.
- Borthakur G, Ravandi F, Patel K, et al: Retrospective comparison of survival and responses to fludarabine, cytarabine, GCSF (FLAG) in combination with gemtuzumab ozogamicin (GO) or idarubicin (IDA) in patients with newly diagnosed core binding factor (CBF) acute myelogenous leukemia: MD Anderson experience in 174 patients. Am J Hematol 97:1427‑1434, 2022.
- Zhang K, Zhang X, Xu Y, et al: Efficacy of venetoclax combined with hypomethylating agents in young, and unfit patients with newly diagnosed core binding factor acute myeloid leukemia. Blood Cancer J 13:155, 2023.
- Druker BJ, Talpaz M, Resta DJ, et al: Efficacy and safety of a specific inhibitor of the BCR‑ABL tyrosine kinase in chronic myeloid leukemia. N Engl J Med 344:1031‑1037, 2001.
- Kantarjian H, Breccia M, Haddad FG, et al: Management of chronic myeloid leukemia in 2025. Cancer 131:e35953, 2025.
- Kantarjian H, Jabbour E: Chronic myeloid leukemia. In: Longo DL, Fauci AS, Kasper DL, et al (eds): Harrison’s Principles of Internal Medicine, 22nd edition. New York, NY: McGraw-Hill Education; 2026.
- Jabbour E, Kantarjian H: Chronic myeloid leukemia: A review. JAMA 333:1618‑1629, 2025.
- Jabbour E, Kantarjian H: Chronic myeloid leukemia: 2025 update on diagnosis, therapy, and monitoring. Am J Hematol 99:2191‑2212, 2024.
- Haddad FG, Kantarjian H: Navigating the management of chronic phase CML in the era of generic BCR::ABL1 tyrosine kinase inhibitors. J Natl Compr Canc Netw 22:e237116, 2024.
- Kantarjian HM, Welch MA, Jabbour E: Revisiting six established practices in the treatment of chronic myeloid leukaemia. Lancet Haematol 10:e860‑e864, 2023.
- Senapati J, Sasaki K, Issa GC, et al: Management of chronic myeloid leukemia in 2023 – common ground and common sense. Blood Cancer J 13:58, 2023.
- Haddad FG, Jabbour E, Breccia M, et al: Choosing the best frontline BCR::ABL1 tyrosine kinase inhibitor in chronic myeloid leukemia—how to define the treatment value?. Clin Lymphoma Myeloma Leuk 26:57‑65, 2026.
- Keating MJ, O’Brien S, Albitar M, et al: Early results of a chemoimmunotherapy regimen of fludarabine, cyclophosphamide, and rituximab as initial therapy for chronic lymphocytic leukemia. J Clin Oncol 23:4079‑4088, 2005.
- Sharman JP, Egyed M, Jurczak W, et al: Acalabrutinib with or without obinutuzumab versus chlorambucil and obinutuzumab for treatment-naive chronic lymphocytic leukaemia (ELEVATE-TN): A randomised, controlled, phase 3 trial. Lancet 395:1278‑1291, 2020.
- Tam CS, Robak T, Ghia P, et al: Zanubrutinib monotherapy for patients with treatment-naïve chronic lymphocytic leukemia and 17p deletion. Haematologica 106:2354‑2363, 2021.
- Munir T, Cairns DA, Bloor A, et al: Chronic lymphocytic leukemia therapy guided by measurable residual disease. N Engl J Med 390:326‑337, 2024.
- Wierda WG, Jacobs R, Barr PM, et al: Outcomes in high-risk subgroups after fixed-duration ibrutinib + venetoclax for chronic lymphocytic leukemia (CLL)/small lymphocytic lymphoma (SLL): Up to 5.5 years of follow-up in the phase 2 CAPTIVATE study. J Clin Oncol 42(suppl):7009, 2024.
- Jain N, Keating M, Thompson P, et al: Ibrutinib and venetoclax for first-line treatment of CLL. N Engl J Med 380:2095‑2103, 2019.
- Jain N, Keating M, Thompson P, et al: Ibrutinib plus venetoclax for first-line treatment of chronic lymphocytic leukemia: A nonrandomized phase 2 trial. JAMA Oncol 7:1213‑1219, 2021.
- Jain N, Keating MJ, Thompson PA, et al: Combined ibrutinib and venetoclax for first-line treatment of patients with chronic lymphocytic leukemia (CLL): 5-year follow-up data. Blood 142:4635, 2023.
- Jain N, Ferrajoli A, Swaminathan M, et al: Combined pirtobrutinib, venetoclax, and obinutuzumab as first-line treatment of patients with chronic lymphocytic leukemia (CLL). Blood 144:1011, 2024.
- Kantarjian H, Short NJ, Haddad FG, et al: Results of the simultaneous combination of ponatinib and blinatumomab in Philadelphia chromosome-positive ALL. J Clin Oncol 42:4246‑4251, 2024.
- Chiaretti S, Di Trani M, Skert C, et al: First results of the phase III GIMEMA ALL2820 trial comparing ponatinib plus blinatumomab to imatinib and chemotherapy for newly diagnosed adult Ph+ acute lymphoblastic leukemia patients. Blood 146(suppl 1):439, 2025.
- Topp MS, Gökbuget N, Stein AS, et al: Safety and activity of blinatumomab for adult patients with relapsed or refractory B-precursor acute lymphoblastic leukaemia: A multicentre, single-arm, phase 2 study. Lancet Oncol 16:57‑66, 2015.
- Kantarjian H, Thomas D, Jorgensen J, et al: Inotuzumab ozogamicin, an anti-CD22–calecheamicin conjugate, for refractory and relapsed acute lymphocytic leukaemia: A phase 2 study. Lancet Oncol 13:403‑411, 2012.
- Kantarjian H, Thomas D, Jorgensen J, et al: Results of inotuzumab ozogamicin, a CD22 monoclonal antibody, in refractory and relapsed acute lymphocytic leukemia. Cancer 119:2728‑2736, 2013.
- Roddie C, Sandhu KS, Tholouli E, et al: Obecabtagene autoleucel in adults with B-cell acute lymphoblastic leukemia. N Engl J Med 391:2219‑2230, 2024.
- Kantarjian H, Short NJ, Jain N, et al: Hyper-CVAD and sequential blinatumomab without and with inotuzumab in young adults with newly diagnosed Philadelphia chromosome-negative B-cell acute lymphoblastic leukemia. Am J Hematol 100:402‑407, 2025.
- Jabbour E, Lussana F, Martínez-Sánchez P, et al: Subcutaneous blinatumomab in adults with relapsed or refractory B-cell acute lymphoblastic leukaemia: Post-hoc safety and activity analysis from a multicentre, single-arm, phase 1/2 trial. Lancet Haematol 12:e529‑e541, 2025.
- Jabbour E, Short N, Jain N, et al: ALL-808 very promising results of the dose dense (D-D) mini-hyper-CVD-inotuzumab-blinatumomab phase 2 trial in patients with relapsed-refractory acute lymphoblastic leukemia. Clin Lymphoma Myeloma Leuk 24(suppl 1):S284‑S285, 2024.
- Geyer MB, Hsu M, Devlin SM, et al: Overall survival among older US adults with ALL remains low despite modest improvement since 1980: SEER analysis. Blood 129:1878‑1881, 2017.
- Jabbour E, Short NJ, Senapati J, et al: Mini-hyper-CVD plus inotuzumab ozogamicin, with or without blinatumomab, in the subgroup of older patients with newly diagnosed Philadelphia chromosome-negative B-cell acute lymphocytic leukaemia: Long-term results of an open-label phase 2 trial. Lancet Haematol 10:e433‑e444, 2023.
- Stelljes M, Raffel S, Alakel N, et al: Inotuzumab ozogamicin as induction therapy for patients older than 55 years with Philadelphia chromosome–negative B-precursor ALL. J Clin Oncol 42:273‑282, 2024.
- Chevallier P, Leguay T, Kim R, et al: Fractionated inotuzumab ozogamicin combined with low-intensity chemotherapy in older patients with newly diagnosed CD22+ Philadelphia chromosome (Ph)-negative B-cell precursor (BCP) acute lymphoblastic leukemia (ALL): Results of the EWALL-INO study. Blood 140(suppl 1):6114‑6116, 2022.
- Iftikhar A, Sohail S, Rehman MEU, et al: Predictors of early mortality in acute myeloid leukemia: A population based analysis using SEER data. Blood 146(suppl 1):1632, 2025.
- Fernandez HF, Sun Z, Yao X, et al: Anthracycline dose intensification in acute myeloid leukemia. N Engl J Med 361:1249‑1259, 2009.
- Löwenberg B, Ossenkoppele GJ, van Putten W, et al: High-dose daunorubicin in older patients with acute myeloid leukemia. N Engl J Med 361:1235‑1248, 2009.
- DiNardo CD, Jen W-Y, Takahashi K, et al: Long term results of venetoclax combined with FLAG-IDA induction and consolidation for newly diagnosed and relapsed or refractory acute myeloid leukemia. Leukemia 39:854‑863, 2025.
- Bouligny IM, Kantarjian HM, Yilmaz M, et al: A phase II trial of venetoclax in combination with cladribine, idarubicin, and cytarabine in newly diagnosed acute myeloid leukemia and high-risk myelodysplastic syndrome. Blood 144(suppl 1):734, 2024.
- Wang H, Mao L, Yang M, et al: Venetoclax plus 3 + 7 daunorubicin and cytarabine chemotherapy as first-line treatment for adults with acute myeloid leukaemia: A multicentre, single-arm, phase 2 trial. Lancet Haematol 9:e415‑e424, 2022.
- Chua CC, Roberts AW, Reynolds J, et al: Chemotherapy and venetoclax in elderly acute myeloid leukemia trial (CAVEAT): A phase Ib dose-escalation study of venetoclax combined with modified intensive chemotherapy. J Clin Oncol 38:3506‑3517, 2020.
- Goulart H, Kantarjian HM, Daver N, et al: A phase II study of cladribine, idarubicin, and cytarabine (CLIA) in combination with gilteritinib for patients with newly diagnosed FLT3-mutated acute myeloid leukemia. Blood 144(suppl 1):2901, 2024.
- Kantarjian H, Borthakur G, Daver N, et al: Current status and research directions in acute myeloid leukemia. Blood Cancer J 14:163, 2024.
- Kantarjian HM, DiNardo CD, Kadia TM, et al: Acute myeloid leukemia management and research in 2025. CA Cancer J Clin 75:46‑67, 2025.
- Montesinos P, Rodríguez-Veiga R, Bergua JM, et al: Quizartinib for newly diagnosed FLT3-internal tandem duplication–negative AML: The randomized, double-blind, placebo-controlled, phase II QUIWI study. J Clin Oncol 44:42‑53, 2026.
- Stein EM, DiNardo CD, Fathi AT, et al: Ivosidenib or enasidenib combined with intensive chemotherapy in patients with newly diagnosed AML: A phase 1 study. Blood 137:1792‑1803, 2021.
- Pratz KW, Jonas BA, Pullarkat V, et al: Long-term follow-up of VIALE-A: Venetoclax and azacitidine in chemotherapy-ineligible untreated acute myeloid leukemia. Am J Hematol 99:615‑624, 2024.
- Jen W-Y, Lachowiez CA, Marvin-Peek J, et al: Optimizing lower intensity triplet therapy in acute myeloid leukemia: A practical guide. Blood Cancer J 16:6, 2025.
- Kadia TM, Reville PK, Wang X, et al: Phase II study of venetoclax added to cladribine plus low-dose cytarabine alternating with 5-azacitidine in older patients with newly diagnosed acute myeloid leukemia. J Clin Oncol 40:3848‑3857, 2022.
- DiNardo CD, Marvin-Peek J, Loghavi S, et al: Outcomes of frontline triplet regimens with a hypomethylating agent, venetoclax, and isocitrate dehydrogenase inhibitor for intensive chemotherapy–ineligible patients with isocitrate dehydrogenase–mutated AML. J Clin Oncol 43:2692‑2699, 2025.
- Short NJ, Daver N, Dinardo CD, et al: Azacitidine, venetoclax, and gilteritinib in newly diagnosed and relapsed or refractory FLT3-mutated AML. J Clin Oncol 42:1499‑1508, 2024.
- Issa GC, Cuglievan B, Daver N, et al: Phase I/II study of the all-oral combination of revumenib (SNDX-5613) with decitabine/cedazuridine (ASTX727) and venetoclax (SAVE) in R/R AML. Blood 144(suppl 1):216, 2024.
- Zeidner JF, Lin TL, Welkie RL, et al: Azacitidine, venetoclax, and revumenib for newly diagnosed NPM1-mutated or KMT2A-rearranged AML. J Clin Oncol 43:2606-2615, 2025.
- Freireich EJ, Wiernik PH, Steensma DP: The leukemias: A half‑century of discovery. J Clin Oncol 32:3463‑3469, 2014.
*Author’s note: Emil J. Freireich, MD, FASCO, died on February 1, 2021. I am indebted to him for encouraging me to question, challenge, and overturn established dogmas and traditions in leukemia care.
Dr. Kantarjian is a professor in the Department of Leukemia at The University of Texas MD Anderson Cancer Center in Houston. Dr. Freireich (deceased) led the Leukemia Research Program at The University of Texas MD Anderson Cancer Center.

