Complete lymph node dissection did not improve survival in melanoma patients randomized to this practice, vs sentinel lymph node biopsy alone, German investigators reported at the 2015 ASCO Annual Meeting.1
“This is the first study that tested the typical recommendation of complete lymph node dissection in patients with positive sentinel nodes, and we cannot confirm this recommendation,” said senior study author Claus Garbe, MD, Professor of Dermatology at the University of Tübingen, Germany, speaking at a press briefing.
The multicenter DeCOG study found no significant treatment-related differences in 5-year recurrence-free survival, distant metastases-free survival, and melanoma-specific survival between the complete lymph node dissection and observation arms, first study author Ulrike Leiter, MD, also of the University of Tübingen, reported at an ASCO session.
“The group with complete lymphadenectomy showed better disease control in the regional lymph node basin, but completed lymphadenectomy was not associated with improved distant metastases-free survival, recurrence-free survival, or melanoma-specific survival,” said Dr. Leiter. “Based on our findings, complete lymphadenectomy cannot be recommended in melanoma patients with micrometastases.”
DeCOG Details
Investigators screened 1,269 patients who had stage III cutaneous melanoma of the trunk and extremities and positive sentinel lymph node biopsy. All patients had micrometastases but no palpable nodes.
Of them, 473 patients ultimately agreed to randomization and were included in the intent-to-treat analysis: 233 in the observation arm and 240 in the complete lymph node dissection arm. Patients had a median tumor thickness of 2.4 mm, and about 40% of tumors were ulcerated. Both cohorts underwent close monitoring for 3 years, including lymph node ultrasonography every 3 months and computed tomography/magnetic resonance imaging or positron-emission tomography scans every 6 months.
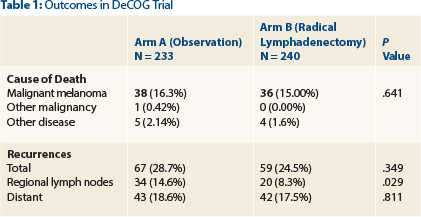
No statistically significant differences were observed in the primary endpoint—distant metastases-free survival—at 80.4% in the observation arm and 80.1% in the complete lymph node dissection arm (hazard ratio = 1.02, P = .92). The only significant difference was observed for regional recurrences (Table 1).
“The radical lymphadenectomy arm had less metastases in the regional nodal basin, which was the only statistically significant difference,” she noted.
Poor prognostic factors included > 1 mm cancer in the sentinel lymph node biopsy sample (hazard ratio = 2.37, P < .0001) and tumor thickness > 2 mm (hazard ratio = 2.36, P = .007).
Dr. Leiter emphasized that accrual to the study was difficult—only one of every three patients agreed to be randomized—and therefore the study population was smaller than designated by the study design. “It was powered to detect a difference of 10% [in the primary endpoint], and to date, that difference in both arms was only 1.1% and the difference in the 3-year distant metastasis free survival was 0.3% (80.4% vs 80.1%).” The investigators will conduct another analysis in 3 years. ■
Disclosure: Drs. Leiter and Garbe reported no potential conflicts of interest.
Reference
1. Leiter U, Stadler R, Mauch C, et al: Survival of SLNB-positive melanoma patients with and without complete lymph node dissection: A multicenter, randomized DeCOG trial. 2015 ASCO Annual Meeting. Abstract LBA9002. Presented May 30, 2015.