The third-generation tyrosine kinase inhibitor ponatinib showed robust efficacy in the 10-month follow-up of the phase II PACE trial (Ponatinib Ph+ALL and CML Evaluation), which is evaluating ponatinib in treatment-refractory chronic myeloid leukemia (CML)1 At the 2012 ASCO Annual Meeting, Jorge E. Cortes, MD, of The University of Texas MD Anderson Cancer Center, Houston, reported, “Responses occurred early and were durable. In chronic-phase patients, 93% are projected to remain in a molecular cytogenetic response at 1 year.”
Overcomes T315I mutation
PACE is an open-label study that enrolled patients with CML or Philadelphia chromosome–positive acute lymphoblastic leukemia who had previously been treated with, and proved resistant or intolerant to, nilotinib (Tasigna) or dasatinib (Sprycel), or who had the T315I mutation (N = 499). Ponatinib was designed to overcome the difficult-to-treat T315I mutation, which it showed capable of doing in this study.
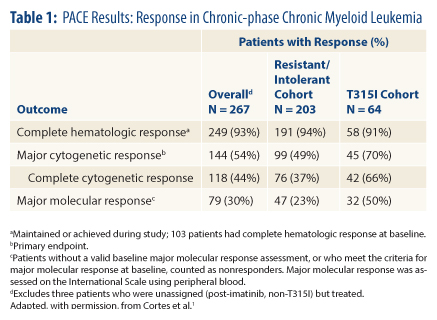
Chronic-phase patients have now been followed for a median of 10 months and blast-phase patients for 6 months. Molecular cytogenetic response, the primary endpoint, was achieved by 54% of chronic-phase patients, including 70% of patients with the T315I mutation and 49% of patients who were refractory/intolerant to dasatinib or nilotinib.
In the accelerated phase, the primary endpoint of major hematologic response was achieved by 58%, including 60% refractory/intolerant and 50% with T315I mutation. In blast-phase CML, a major hematologic response was reached by 34%, 35%, and 33%, respectively.
Describing the results in chronic-phase CML patients in more detail, Dr. Cortes reported that “the majority of the cytogenetic responses are complete cytogenetic responses, and we also see a high rate of major molecular responses (Table 1, page 28).”
Effect of Time and Prior Treatment
Responses improved over time. While at 3 months, 38% of all patients achieved molecular cytogenetic response, this increased to 49% at 6 months and to 53% at 9 months, and the trend was observed in all cohorts. A major molecular response was achieved by 13%, 24%, and 28% of patients at these respective time points.
Some correlation was observed between number of prior treatments and response, with best responses seen in patients who had received no more than two fewer tyrosine kinase inhibitors. But even in those who received three tyrosine kinase inhibitors, rates of complete cytogenetic response were 34% in the resistant/intolerant cohort and 48% with the T315I mutation.
“These results demonstrate that ponatinib is very effective against CML in this very heavily pretreated cohort. Responses occurred regardless of mutation status or disease stage and they improved over time,” he said. A study of ponatinib as initial treatment is being initiated.
Ponatinib ‘Outpacing’ Other Agents
Michael J. Mauro, MD, of Oregon Health and Science University, Portland, the invited discussant of the abstract, commented that while the data are still early, “Ponatinib in the third line is matching or ‘outpacing’ response rates we saw in the second line with nilotinib and dasatinib. Our third-line treatment is proving better than our second-line treatment.”
Cytogenetic and molecular responses were high, irrespective of the number of prior tyrosine kinase inhibitors administered, and were consistently impressive in the less-treated T315I cohort. “But some loss of cytogenetic response was noted with increasing exposure [in this group] and in the subset of patients with T315I and additional mutations,” he said. “This may be proof that we do want to treat earlier.”
With ponatinib moving toward FDA approval, Dr. Mauro predicted how the drug might best be used. “It may be ‘built’ for T315I patients, which gives justification for mutation analysis in resistant CML to find this subset,” he said. “And its performance overall and durability of response justify approval and rapid implementation for better salvage.”
Certainly for the subset of patients with evidence of early treatment resistance, ponatinib “may be the next step,” he said. Perhaps patients with T315I mutations who are resistant to imatinib (Gleevec) should be introduced earlier to the drug.
“Ponatinib is revolutionizing CML and providing the remarkable ability to salvage resistant disease, especially T315I patients,” he concluded. “Its earlier use may prove even better.” ■
Disclosure: Dr. Cortes serves in an unpaid consulting or advisory role for ARIAD, Bristol-Myers Squibb, ChemGenex, Novartis, and Pfizer, and has received research funding from ARIAD, Bristol-Myers Squibb, ChemGenex, Deciphera, Novartis, and Pfizer. Dr. Mauro has been consultant or advisor to and received honoraria from Bristol-Myers Squibb and Novartis.
Reference
1. Cortes JE, Kim D, Pinilla-Ibarz J, et al: PACE: A pivotal phase II trial of ponatinib in patients with CML and Ph+ALL resistant or intolerant to dasatinib or nilotinib, or with the T315I mutation. 2010 ASCO Annual Meeting. Abstract 6503. Presented June 4, 2012.