IN THE PADDY TRIAL, involving more than 10,000 women with early invasive breast cancer, the presence of disseminated tumor cells at diagnosis or primary surgery was an independent prognostic factor for overall, disease-free, and distant disease–free survival. The study findings were presented at the 2018 San Antonio Breast Cancer Symposium by Andreas D. Hartkopf, MD, of the University of Tübingen, Germany.1

Andreas D. Hartkopf, MD
“It has been previously shown that metastatic spread derives from early breast cancer lesions. In this study, disseminated tumor cell detection was an independent factor for metastatic relapse and poor overall survival, and its impact on metastatic relapse was most pronounced in luminal B patients,” Dr. Hartkopf said.
The PADDY trial was a pooled analysis of the impact of prognosis in early breast cancer. Of 10,307 patients, disseminated tumor cells were detected in the bone marrow of 2,814 patients (27.3%). Their detection was associated with local tumor burden as well as biologic subtype and had prognostic significance, Dr. Hartkopf reported.
“Disseminated tumor cell detection was an independent factor for metastatic relapse and poor overall survival, and its impact on metastatic relapse was most pronounced in luminal B [breast cancer].”— Andreas D. Hartkopf, MD
Tweet this quote
PADDY Design
THE STUDY included 10,811 patients (10,307 evaluable) with early invasive breast cancer treated at 11 centers (mostly in Europe) between 1986 and 2017. All had bone marrow aspiration performed at the time of primary diagnosis or primary surgery and had not received systemic treatment. Patients were defined as being disseminated tumor cell–positive if at least one tumor cell was detected by cytokeratin staining. Survival times were measured from the time of bone marrow sampling.
Of the 10,307 patients, 27.3% were defined as disseminated tumor cell–positive. A number of baseline characteristics were associated with positivity, including grade, tumor size, nodal status, hormone receptor status, and HER2 status, as follows (all P < .001):
- Grade 3 (29.9%), grade 2 (27.2%), grade 1 (21.2%)
- T4 (45.7%), T3 (37.0%), T2 (29.7%), T1 (24.7%)
- Node-positive (31.2%), node-negative (24.4%)
- Estrogen receptor–negative (30.2%), estrogen receptor–positive (24.8%)
- Progesterone receptor–negative (30.1%), progesterone receptor– positive (24.3%)
- HER2-positive (29.6%), HER2-negative (23.2%).
Prognostic Almost Across the Board
THE COX regression model for the multivariate analysis included age, menopausal status, histology, tumor size, nodal status, biologic subtype, and disseminated tumor cells. Disseminated tumor cells proved to be prognostic for overall, disease-free, and distant disease–free survival (Table 1).
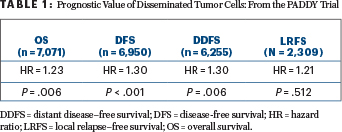
The HER2 biologic subtype was the most likely to be disseminated tumor cell–positive (29.6%), but the only statistically significant interaction between disseminated tumor cells and biologic subtype was for luminal B breast cancer. In that subgroup, the hazard ratio for distant disease–free survival was 2.34 (95% confidence interval [CI] = 1.60–3.43). For the HER2 subtype, the hazard ratio was 1.79 (95% CI = 1.29–2.50). There was no statistically significant interaction between the presence of disseminated tumor cells and overall survival, he reported.
Dr. Hartkopf added that disseminated tumor cells were prognostic almost across the board. “We have previously reported that we can find disseminated tumor cells even in patients with in situ carcinoma, and it impacts survival,” he said.
Remaining Questions
IN RESPONSE to questions from the audience, Dr. Hartkopf said that the use of bisphosphonates in early breast cancer has been shown to have a greater impact on disseminated tumor cell–positive patients, but in the PADDY study, their impact would not be clear. “The decision to use bisphosphonates was partly driven by the detection of disseminated tumor cells, so it would be difficult to show they are predictive,” he said.
The study also does not have information about the median number of disseminated tumor cells and their relative prognostic value, since centers were required to report only disseminated tumor cell positivity. The investigators have such information from some participating sites, however, and may explore this question later, he added. ■
DISCLOSURE: Dr. Hartkopf has received personal fees from AstraZeneca, Celgene, Daiichi Sankyo, Eisai, Genomic Health, Lilly, Merck & Co., Novartis, Pfizer, Roche, Tesaro, and Teva.
REFERENCE
1. Hartkopf AD, Brucker SY, Taran F-A, et al: International pooled analysis of the prognostic impact of disseminated tumor cells from the bone marrow in early breast cancer: Results from the PADDY study. 2018 San Antonio Breast Cancer Symposium. Abstract GS5-07. Presented December 7, 2018.


