In the storied English fable “Goldilocks and the Three Bears,” a child enters a home and tastes three bowls of porridge. She prefers a bowl that is not too cold and not too hot but one that has just the right temperature. This “Goldilocks principle” of “just the right amount” can be applied to the discussion of the postoperative management of prostate cancer.

Manuj Agarwal, MD
Putting Trial Data Into Context
Three simultaneous trials1-3 and a planned meta-analysis4 recently provided the only level I data in this context. The RADICALS,1 RAVES,2 and GETUG3 trials collectively randomly assigned more than 2,000 patients to receive adjuvant vs salvage radiotherapy after radical prostatectomy. The 5-year event-free survival was nearly identical at 89% vs 88%. One-third to one-half of men could avoid radiotherapy and, in doing so, could avoid the toxicities associated with salvage radiation. For these men, the porridge is too hot!
The conclusion drawn by the authors and adopted by the larger prostate cancer community was that there is no role for routine adjuvant radiation. There should be an asterisk added to this remark that amends it to read: “There is no role for routine adjuvant radiation in the patient cohort represented in these studies.” Men with adverse features were underrepresented, with Gleason score ≥ 8 representing 9% to 17%, pT3b representing 18% to 21%, and pT4a representing 1% of patients. Patients with pathologic node-positive disease were excluded from RAVES and GETUG and represented 5% of the RADICALS cohort. The trials individually and collectively did not report outcomes for high-risk combination subgroups.
These adverse features are associated with a higher risk for biochemical failure, distant metastasis, and cancer-specific mortality,5 and these patients represent a higher-risk cohort. Registry data support that such patients are routinely referred for early salvage radiotherapy.6-8 When they are sent, they present with higher-than-desirable prostate-specific antigen (PSA) values. In one series, 43% of patients who received salvage radiotherapy did so after reaching a value of 0.5 ng/mL.6 This is unfortunate, as the initiation of salvage radiotherapy at a higher PSA value has been associated with increased rates of biochemical failure and distant metastases.9 By routinely withholding radiotherapy, we run the risk of offering porridge that is too cold.
Search for Optimal Therapy for Men With Adverse Pathology
SWOG S8794, EORTC 22911, and ARO 96-0210-12 represent the available prospective data on investigation of salvage radiotherapy. They are dated and fraught with flaws, including a large proportion of patients with PSA disease progression. We are left with a hole that only big data can fill. In a study recently reported in the Journal of Clinical Oncology—and summarized in this issue of The ASCO Post—Tilki et al sought to investigate the optimal therapy for men with high-risk adverse features.13
In the study, they retrospectively reviewed prospectively collected data from approximately 26,000 patients from 5 institutions with pT2–4N0 or N1M0 disease; approximately 3% of the total cohort received adjuvant radiotherapy, and approximately 18% received early salvage radiotherapy. Among a total of 2,424 patients with adverse pathology, when those with pN1 disease were included, 428 received adjuvant radiotherapy, 965 received no radiotherapy, and 1,031 received salvage radiotherapy; among a total of 933 with adverse pathology, when those with pN1 disease were excluded, 109 received adjuvant radiotherapy, 379 had no radiotherapy, and 445 received salvage radiotherapy.
After a median follow-up of approximately 8 years, adjuvant radiotherapy was associated with a reduction in all-cause-mortality vs salvage radiotherapy in men with adverse pathology, whether they had pN0 or pN1 disease. There was no reduction in all-cause mortality with adjuvant radiotherapy in men without adverse pathology.
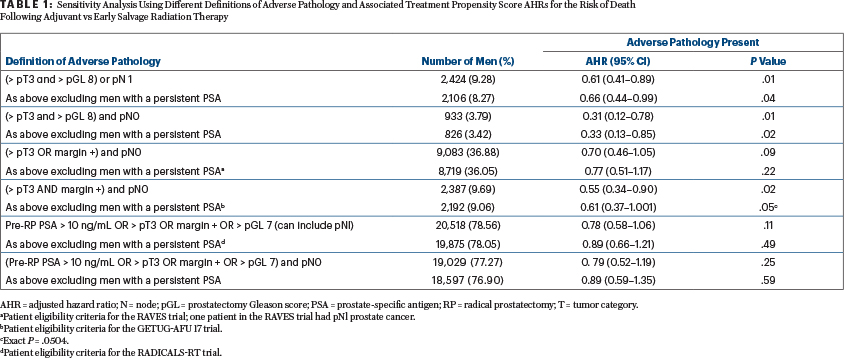
They further performed a sensitivity analysis using different combinations of adverse features, as depicted in Table 1 below—a reproduction of Table 3 from the Tilki et al article. As can be seen, men with ≥ T3 and ≥ Gleason 8 benefited from adjuvant radiotherapy regardless of nodal involvement and/or a persistent PSA level, as did men with ≥ T3, pN0 disease with a positive margin and a persistent PSA level. Note that men with no single adverse feature benefited from adjuvant radiotherapy. The inclusion criteria for the prospective RAVES, GETUG, and RADICALS trials previously discussed are indicated by the superscripts a, b, and d at the bottom of Table 1. As expected, these patients did not have a benefit from adjuvant radiotherapy.
Similar conclusions were found in a prior retrospective multi-institutional analysis.14 Approximately 500 men with adverse features after prostatectomy and with genomic data were reviewed. The genomic classifier Decipher was used,* which has the potential to prognosticate the risk of recurrence and metastases after surgery.15 They found that pT3b, ≥ Gleason score 8, pN+, and a high Decipher score (> 0.6) were independently predictive of recurrence, and the combination of any of these two factors was associated with a 10-year recurrence rate of 10% with adjuvant radiotherapy vs 40% with initial observation. (*The Decipher test looks at the activity of 22 genes in prostate tumors and calculates a score. A low Decipher score of between 0 and 0.45 suggests a lower risk of metastasis and death from prostate cancer, whereas intermediate Decipher scores of between 0.45 and 0.6 as well as high Decipher scores of between 0.6 and 1 suggest a higher risk of metastasis and death from prostate cancer.)
Risk-Adapted Strategy for Selecting Patients for Treatment
In the era of individualized medicine, adopting a “one-size-fits-all” approach may place our patients at higher risk for recurrence and death. By adopting a risk-adapted strategy, informed by data supporting early salvage for low-risk patients and adjuvant therapy for high-risk patients, we can apply a treatment that is “just right” for the individual patient sitting across from us. It may be prudent for high-risk patients to be routinely offered consultation with a radiation oncologist.
The study by Tilki et al is restricted by the usual limitations of retrospective analyses. These retrospective data, as well as the available prospective data, include patients who were treated in the era before metabolic imaging, which in the salvage setting has been shown to alter radiotherapy decision-making and improve biochemical outcomes.16 There remain questions regarding radiation volumes, radiation dose, radiation technique, and the use of androgen-deprivation therapy.
Dr. Agarwal is Assistant Professor of Radiation Oncology at the University of Pennsylvania and Medical Director of Radiation Oncology at the Abramson Cancer Center at Penn Presbyterian Medical Center, Philadelphia.
DISCLOSURE: Dr. Agarwal reported no conflicts of interest.
REFERENCES
1. Parker CC, Clarke NW, Cook AD, et al: Timing of radiotherapy after radical prostatectomy (RADICALS-RT): A randomised, controlled phase 3 trial. Lancet 396:1413-1421, 2020.
2. Kneebone A, Fraser-Browne C, Duchesne GM, et al: Adjuvant radiotherapy versus early salvage radiotherapy following radical prostatectomy (TROG08.03/ANZUP RAVES): A randomised, controlled, phase 3, non-inferiority trial. Lancet Oncol 21:1331-1340, 2020.
3. Carrie C, Magné N, Burban-Provost P, et al: Short-term androgen deprivation therapy combined with radiotherapy as salvage treatment after radical prostatectomy for prostate cancer (GETUG-AFU 16): A 112-month follow-up of a phase 3, randomised trial. Lancet Oncol 20:1740-1749, 2019.
4. Vale CL, Fisher D, Kneebone A, et al: Adjuvant or early salvage radiotherapy for the treatment of localised and locally advanced prostate cancer: A prospectively planned systematic review and meta-analysis of aggregate data. Lancet 396:1422-1431, 2020.
5. Tilki D, Mandel P, Schlomm T, et al: External validation of the CAPRA-S score to predict biochemical recurrence, metastasis and mortality after radical prostatectomy in a European cohort. J Urol 193:1970-1975, 2015.
6. Hawken SR, Spratt DE, Qi J, et al: Utilization of salvage radiation therapy for biochemical recurrence after radical prostatectomy. Int J Radiat Oncol Biol Phys 104:1030-1034, 2019.
7. Morgan TM, Hawken SR, Ghani KR, et al: Variation in the use of postoperative radiotherapy among high-risk patients following radical prostatectomy. Prostate Cancer Prostatic Dis 19:216-221, 2016.
8. Kalbasi A, Swisher-McClure S, Mitra N, et al: Low rates of adjuvant radiation in patients with nonmetastatic prostate cancer with high-risk pathologic features. Cancer 120:3089-3096, 2014.
9. Tendulkar RD, Agrawal S, Gao T, et al: Contemporary update of a multi-institutional predictive nomogram for salvage radiotherapy after radical prostatectomy. J Clin Oncol 34:3648-3654, 2016.
10. Thompson IM, Tangen CM, Paradelo J, et al: Adjuvant radiotherapy for pathological T3N0M0 prostate cancer significantly reduces risk of metastases and improves survival: Long-term follow up of a randomized clinical trial. J Urol 181:956-962, 2009.
11. Bolla M, van Poppel H, Tombal B, et al: Postoperative radiotherapy after radical prostatectomy for high-risk prostate cancer: Long-term results of a randomised controlled trial (EORTC trial 22911). Lancet 380:2018-2027, 2012.
12. Wiegel T, Bartkowiak D, Bottke D, et al: Adjuvant radiotherapy versus wait-and-see after radical prostatectomy: 10-year follow-up of the ARO 96-02/AUO AP 09/95 trial. Eur Urol 66:243-250, 2014.
13. Tilki D, Chen MH, Wu J, et al: Adjuvant versus early salvage radiation therapy for men at high risk for recurrence following radical prostatectomy for prostate cancer and the risk of death. J Clin Oncol 39:2284-2293, 2021.
14. Den RB, Yousefi K, Trabulsi EJ, et al: Genomic classifier identifies men with adverse pathology after radical prostatectomy who benefit from adjuvant radiation therapy. J Clin Oncol 33:944-951, 2015.
15. Dalela D, Santiago-Jiménez M, Yousefi K, et al: Genomic classifier augments the role of pathological features in identifying optimal candidates for adjuvant radiation therapy in patients with prostate cancer: Development and internal validation of a multivariable prognostic model. J Clin Oncol 35:1982-1990, 2017.
16. Jani AB, Schreibmann E, Goyal S, et al: 18 F-fluciclovine-PET/CT imaging versus conventional imaging alone to guide postprostatectomy salvage radiotherapy for prostate cancer (EMPIRE-1): A single centre, open-label, phase 2/3 randomised controlled trial. Lancet 397:1895-1904, 2021.

